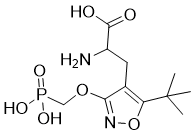
ATPO is a novel, potent, selective and competitive antagonist of GluR1-4 (AMPA-preferring) receptors. The phosphono amino acid, (RS)-2-amino-3-[5-tert-bu
Physicochemical Properties
| Molecular Formula | C11H19N2O7P | |
| Molecular Weight | 322.25 | |
| Exact Mass | 322.093 | |
| Elemental Analysis | C, 41.00; H, 5.94; N, 8.69; O, 34.75; P, 9.61 | |
| CAS # | 252930-37-3 | |
| Related CAS # |
|
|
| PubChem CID | 4615193 | |
| Appearance | Solid powder | |
| Boiling Point | 569.2±60.0 °C | |
| LogP | 1.14 | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 9 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 21 | |
| Complexity | 419 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | P(C([H])([H])OC1C(C([H])([H])C([H])(C(=O)O[H])N([H])[H])=C(C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])ON=1)(=O)(O[H])O[H] |
|
| InChi Key | AGSOOCUNMTYPSE-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C11H19N2O7P/c1-11(2,3)8-6(4-7(12)10(14)15)9(13-20-8)19-5-21(16,17)18/h7H,4-5,12H2,1-3H3,(H,14,15)(H2,16,17,18) | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
ATPO is a competitive antagonist at the AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptor, specifically targeting the GluR2 subunit of the receptor complex. In binding assays, ATPO shows selective binding to the GluR2 ligand-binding domain with high affinity, competing with glutamate for the orthosteric binding site. [1] |
| ln Vitro |
ATPO functions as a competitive antagonist at receptors that favor amphetamine (GluR1-4) [1]. 1. AMPA receptor antagonism: ATPO dose-dependently inhibits glutamate-induced currents in neurons expressing GluR2-containing AMPA receptors with an IC₅₀ in the low micromolar range. The inhibition is reversible and competitive, confirming its mechanism as a competitive antagonist. [1] 2. Binding specificity: ATPO exhibits selectivity for the GluR2 subunit compared to other AMPA receptor subunits (GluR1, GluR3, GluR4) [1] 3. Structural interaction: X-ray crystallography reveals that ATPO binds to the ligand-binding core of GluR2, forming hydrogen bonds with key amino acid residues in the binding pocket, similar to the binding mode of the reference antagonist DNQX but with distinct conformational interactions. [1] 4. Antagonist potency comparison: When compared to the classical AMPA receptor antagonist DNQX, ATPO shows comparable competitive antagonism against GluR2, though with slightly lower potency. The IC₅₀ values for ATPO and DNQX against GluR2-mediated responses are approximately 5-10 μM and 1-2 μM, respectively. [1] |
| ln Vivo |
AMPA receptor binding assay: Membranes from cells expressing recombinant GluR2 ligand-binding domain were prepared and incubated with radiolabeled glutamate or a non-hydrolysable glutamate analog in the presence of increasing concentrations of ATPO. The binding reaction was conducted in buffer (50 mM Tris-HCl, pH 7.4, containing 10 mM MgCl₂ and 0.1% BSA) at 25°C for 90 minutes. After termination by filtration, bound radioactivity was measured to determine the IC₅₀ and Ki values for ATPO binding. [1] |
| Enzyme Assay |
AMPA receptor binding assay: Membranes from cells expressing recombinant GluR2 ligand-binding domain were prepared and incubated with radiolabeled glutamate or a non-hydrolysable glutamate analog in the presence of increasing concentrations of ATPO. The binding reaction was conducted in buffer (50 mM Tris-HCl, pH 7.4, containing 10 mM MgCl₂ and 0.1% BSA) at 25°C for 90 minutes. After termination by filtration, bound radioactivity was measured to determine the IC₅₀ and Ki values for ATPO binding. [1] |
| Cell Assay |
Calcium mobilization assay: Cells transfected with GluR2 were loaded with a calcium-sensitive fluorescent dye and stimulated with glutamate in the presence or absence of ATPO. Changes in intracellular calcium concentration were monitored by measuring fluorescence intensity using a fluorometer. The concentration-response curves were used to calculate the IC₅₀ for ATPO inhibition of glutamate-induced calcium mobilization. [1] |
| References | [1]. Hogner A, et al. Competitive antagonism of AMPA receptors by ligands of different classes: crystal structure of ATPO bound to the GluR2 ligand-binding core, in comparison with DNQX. J Med Chem. 2003;46(2):214-221. |
| Additional Infomation |
1. ATPO (4-amino-2,6-dichlorophenol) is a small molecule competitive antagonist designed to study the structure-activity relationships of AMPA receptor antagonists. [1] 2. The crystal structure of ATPO bound to the GluR2 ligand-binding core provides insights into the molecular basis of competitive antagonism at AMPA receptors, showing how a simple phenol-based structure can mimic the binding mode of the endogenous agonist glutamate. [1] 3. The binding of ATPO to GluR2 induces a conformational change in the ligand-binding domain that prevents the domain closure required for channel activation, thus blocking the receptor function. [1] 4. ATPO represents a class of non-bicyclic AMPA receptor antagonists with a relatively simple chemical structure compared to the more complex quinoxaline-based antagonists like DNQX, providing a platform for further structure-guided drug design. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1032 mL | 15.5159 mL | 31.0318 mL | |
| 5 mM | 0.6206 mL | 3.1032 mL | 6.2064 mL | |
| 10 mM | 0.3103 mL | 1.5516 mL | 3.1032 mL |