Physicochemical Properties
| Molecular Formula | C26H22N2O4 |
| Molecular Weight | 426.463886737823 |
| Exact Mass | 426.16 |
| Elemental Analysis | C, 73.23; H, 5.20; N, 6.57; O, 15.01 |
| CAS # | 2089314-64-5 |
| PubChem CID | 126718388 |
| Appearance | Solid powder |
| Density | 1.269±0.06 g/cm3(Predicted) |
| Boiling Point | 703.9±50.0 °C(Predicted) |
| LogP | 4.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 32 |
| Complexity | 627 |
| Defined Atom Stereocenter Count | 0 |
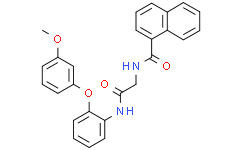
| SMILES | O(C1C=CC=C(C=1)OC)C1=CC=CC=C1NC(CNC(C1=CC=CC2C=CC=CC1=2)=O)=O |
| InChi Key | HDMONPHKMIZXDH-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H22N2O4/c1-31-19-10-7-11-20(16-19)32-24-15-5-4-14-23(24)28-25(29)17-27-26(30)22-13-6-9-18-8-2-3-12-21(18)22/h2-16H,17H2,1H3,(H,27,30)(H,28,29) |
| Chemical Name | N-[2-[2-(3-methoxyphenoxy)anilino]-2-oxoethyl]naphthalene-1-carboxamide |
| Synonyms | NSC789796; NSC 789796; 2089314-64-5; EZ94L3M5SX; N-[2-[[2-(3-methoxyphenoxy)phenyl]amino]-2-oxoethyl]-1-Naphthalenecarboxamide; N-(2-((2-(3-Methoxyphenoxy)phenyl)amino)-2-oxoethyl)-1-naphthalenecarboxamide; RefChem:1090906; AOH1,996; N-(2-((2-(3-Methoxyphenoxy)phenyl)amino)-2-oxoethyl)-1-naphthamide; N-[2-[[2-(3-Methoxyphenoxy)phenyl]amino]-2-oxoethyl]-1-naphthamide; NSC-789796; AOH1996; AOH-1996; AOH 1996; |
| HS Tariff Code | 2934.99.9005 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Proliferating cell nuclear antigen (PCNA), especially the cancer - associated variant of PCNA. [2] |
| ln Vitro |
AOH1996 selectively kills cancer cells. AOH1996 enhances the interaction between PCNA and the largest subunit of RNA polymerase II, RPB1, and dissociates PCNA from actively transcribed chromatin regions, while inducing DNA double-stranded breaks in a transcription-dependent manner. Attenuation of RPB1 interaction with PCNA, by a point mutation in RPB1's PCNA-binding region, confers resistance to AOH1996 [2].
AOH1996 can selectively target PCNA, enhance the interaction between PCNA and RPB1, leading to the overall degradation of RPB1 and the collapse of DNA replication forks in actively transcribed chromatin regions. It can induce cell - cycle arrest (G2/M or S - phase arrest) and apoptosis in cancer cells, but not in non - malignant cells. The drug inhibits the growth of about 50% of tumor cells at a median drug concentration of approximately 300 nM. The research team tested AOH1996 in more than 70 cancer cell lines and several groups of normal control cells, and the results showed that it could prevent cells with damaged DNA from dividing in the G2/M phase and prevent the replication of error - prone DNA in the S phase. [2] |
| ln Vivo |
Orally administrable and metabolically stable, AOH1996 suppresses tumor growth as a monotherapy or as a combination treatment but causes no discernable side effects. Inhibitors of transcription replication conflict resolution may provide a new and unique therapeutic avenue for exploiting this cancer-selective vulnerability [2].
AOH1996 can almost completely inhibit the growth of xenograft tumors without causing any obvious toxicity to experimental animals. It targets the PCNA in cancer cells, disrupts the PCNA - TRC interface in cancer cells, and plays an effective selective anti - cancer role. [2] |
| Enzyme Assay |
Thermal denaturation assay for PCNA-AOH1996 interaction [2] The assay was performed using a BioRad CFX Connect Real-Time PCR Detection System. Protein (PCNA), inhibitors (AOH1996 and AOH1160–1LE), and 200x SYPRO orange dye were diluted into phosphate buffered saline (PBS). The final concentration of recombinant PCNA was 9 μM, and the final compound concentrations were at 0, 10, or 30 μM. Sample plates were heated from 25°C to 95°C with heating increments of 0.5 °C/min. Fluorescence intensity was measured within the excitation/emission ranges 470–505/540–700 nm. Compound metabolism in liver microsome [2] AOH1160 analogs were incubated at 37°C with human liver microsomes in the absence or presence of NADPH. An aliquot of the reaction mixture was then taken after various incubation times, and the Concentration of Compound determined by liquid chromatography–tandem mass spectrometry (LC-MS/MS) as previously described. |
| Cell Assay |
Cell cycle analysis[2] Cells were seeded in a 6-well plate at 1×105/ml. After treatment with the compound, cells were fixed in 60% ethanol and stained with propidium iodide (PI). PI fluorescence intensity within the cells was measured by flow cytometry, and the data was analyzed using the FlowJo program. Cell growth and apoptosis assays[2] Cells were seeded at 5 × 103/ml or 3 × 104/ml into a 96-well plate, depending on the cell lines, and were treated with various concentrations of AOH1996 for 72 h after being allowed to attach overnight. Cell growth was measured by the CellTiter-Glo assay according to the manufacturer’s instruction. Alternatively, cell growth was analyzed by the IncuCyte S3 Live-cell Analysis Systems, which measures cell confluence by periodic imaging. The National Cancer Institute (NCI) examined the effect of AOH1996 on the NCI60 panel of cell lines using the standard 5-dose assay containing sulforhodamine B (SRB). The assay is described in more details at (https://dtp.cancer.gov/discovery_development/nci-60/methodology.htm). The 50% growth inhibition concentration (GI50) was calculated by the NCI. Cell apoptosis was measured on a chamber slide at a seeding density of 1×105/ml. After treatment with 500 nM AOH1996 for 24 h, cells were fixed and analyzed using the terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling (TUNEL) assay containing TMR red in situ cell death detection kit. AOH1996 can almost completely inhibit the growth of xenograft tumors without causing any obvious toxicity to experimental animals. It targets the PCNA in cancer cells, disrupts the PCNA - TRC interface in cancer cells, and plays an effective selective anti - cancer role. [2] |
| Animal Protocol |
Pharmacokinetic (PK) study in animals[2] An oral dosing solution was prepared by dissolving AOH1996 (40 mg) in a mixture of Kolliphor EL (840 mg) and Poloxamer P124 (120 mg). For the mouse study, blood samples were collected from ES1e/SCID mice (3 male and 3 female) per dosing group by cardiac puncture at 10, 20, and 30 min and 1, 2, 4, 6, and 24 h after dosing. For the dog study, blood samples were collected from 3 male beagle dogs per dosing group by venipuncture of peripheral veins at 5, 15, and 30 min and 1, 2, 4, 8, 12, and 24 h after dosing. Following removal of blood cells, plasma concentration of AOH1996 was determined by LC-MS/MS as described.17 Oral PK was determined using standard non-compartmental methods. In vivo tumor model[2] Compounds were tested in the ES1e/SCID mice, originally provided by Dr. Philip M. Potter of the St. Jude Children’s Research Hospital.49 SK-N-BE(2)c and SK-N-AS neuroblastoma cells were suspended in Matrigel at 5 × 107/ml and 4 × 107/ml, respectively, after being harvested and washed twice in PBS. The cell suspension (0.1 mL) was subcutaneously injected into the right flank of each ES1e/SCID mouse. AOH1996 was dosed orally. CPT-11 was given by intraperitoneal injection. Tumor size and animal weight were measured weekly. At the end of the experiment, tumors were isolated from sacrificed mice and analyzed by immunohistochemistry staining with antibodies specific for phosphor-Chk1 and γH2A.X. |
| ADME/Pharmacokinetics |
- Compared with AOH1160, the half - life of AOH1996 in mouse oral pharmacokinetics studies is increased by about 27%, which is more suitable for oral administration. [3] |
| Toxicity/Toxicokinetics |
In pre - clinical studies, AOH1996 did not show obvious toxicity, and there was no report of information such as half - lethal dose, liver and kidney toxicity, drug - drug interaction, and plasma protein binding. [2] |
| References |
[1]. Pharmacological targeting of transcription-replication conflict leads to anti-cancer efficacy with minimal side effects in preclinical models. Cancer Research, 2021, 81(13_Supplement): 1269-1269. [2]. Small molecule targeting of transcription-replication conflict for selective chemotherapy. Cell Chem Biol . 2023 Oct 19;30(10):1235-1247.e6. . |
| Additional Infomation |
AOH1996 is a small - molecule oral drug. PCNA is a central “hub” in the DNA replication/repair pathway and interacts with many other cell pathways. It is one of the non - oncogene proteins necessary for the growth and survival of cancer cells. AOH1996 has entered phase I clinical trials, and the research is expected to last for two years, aiming to determine its maximum tolerated dose and evaluate its preliminary efficacy in adults with solid tumors that do not respond to standard treatment. [2] AOH1996 is a secondary carboxamide resulting from the formal condensation of the carboxy group of N-(naphthalen-1-ylcarbonyl)glycine with the amino group of 2-(3-methoxyphenoxy)aniline. It is a first-in-class small molecule inhibitor of PCNA, a protein essential for DNA replication and repair within tumours. It has a role as an antineoplastic agent, an apoptosis inducer and a PCNA inhibitor. It is an aromatic ether, a naphthalenecarboxamide, a monomethoxybenzene, a secondary carboxamide and a diether. |
Solubility Data
| Solubility (In Vitro) | DMSO: > 10 mM |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3449 mL | 11.7244 mL | 23.4489 mL | |
| 5 mM | 0.4690 mL | 2.3449 mL | 4.6898 mL | |
| 10 mM | 0.2345 mL | 1.1724 mL | 2.3449 mL |