Physicochemical Properties
| Molecular Formula | C25H20N2O3 |
| Molecular Weight | 396.437906265259 |
| Exact Mass | 396.147 |
| CAS # | 2089314-57-6 |
| PubChem CID | 126718383 |
| Appearance | Off-white to light yellow solid powder |
| LogP | 4.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 30 |
| Complexity | 575 |
| Defined Atom Stereocenter Count | 0 |
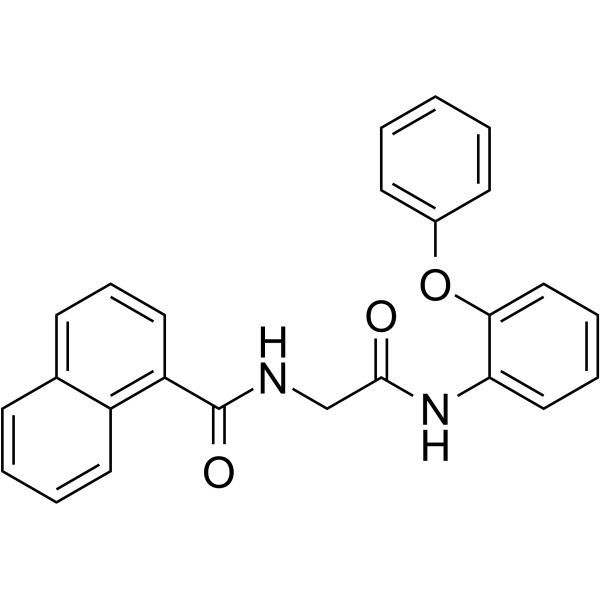
| SMILES | O(C1C=CC=CC=1)C1=CC=CC=C1NC(CNC(C1=CC=CC2C=CC=CC1=2)=O)=O |
| InChi Key | COPRLRLXDBSLET-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H20N2O3/c28-24(17-26-25(29)21-14-8-10-18-9-4-5-13-20(18)21)27-22-15-6-7-16-23(22)30-19-11-2-1-3-12-19/h1-16H,17H2,(H,26,29)(H,27,28) |
| Chemical Name | N-[2-oxo-2-(2-phenoxyanilino)ethyl]naphthalene-1-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The target of AOH1160 is Proliferating Cell Nuclear Antigen (PCNA), a key regulator of DNA replication and repair. Key binding affinity data: - PCNA (human recombinant): Ki = 0.3 μM (ITC assay) [1] |
| ln Vitro |
1. Antiproliferative activity against diverse cancer cell lines: AOH1160 exhibited potent antiproliferative effects on multiple human cancer cell lines (72-hour MTS assay): - Non-small cell lung cancer (A549): IC₅₀ = 0.18 μM [1] - Colorectal cancer (HCT116): IC₅₀ = 0.22 μM [1] - Breast cancer (MCF-7): IC₅₀ = 0.25 μM [1] - Cervical cancer (HeLa): IC₅₀ = 0.15 μM [1] - Chemoresistant colorectal cancer (HCT116/5-FU): IC₅₀ = 0.28 μM [1] - Normal human bronchial epithelial cells (NHBE): IC₅₀ > 5 μM; normal umbilical vein endothelial cells (HUVEC): IC₅₀ > 6 μM, indicating low toxicity to normal cells [1] 2. Inhibition of DNA replication and induction of DNA damage: - EdU incorporation assay: A549 cells treated with AOH1160 (0.1–0.5 μM) for 24 hours showed dose-dependent reduction in DNA synthesis (inhibition rates: 45% at 0.1 μM, 78% at 0.5 μM) [1] - Immunofluorescence staining: 0.3 μM AOH1160 induced a 5.2-fold increase in γ-H2AX foci (DNA double-strand break marker) in HCT116 cells [1] - Western blot: Dose-dependent upregulation of γ-H2AX and p-CHK1 (DNA damage response markers) in A549 and HCT116 cells [1] 3. Cell cycle arrest and apoptosis induction: - Cell cycle analysis (flow cytometry): A549 cells treated with 0.3 μM AOH1160 for 24 hours showed S-phase arrest (58% vs. 32% in vehicle control) [1] - Apoptosis assay (Annexin V/PI staining): HCT116 cells treated with AOH1160 (0.2–1 μM) for 48 hours showed dose-dependent apoptosis (18% at 0.2 μM, 42% at 0.5 μM, 65% at 1 μM) [1] - Western blot: Increased levels of cleaved caspase-3/7/9 and PARP cleavage; decreased anti-apoptotic Bcl-2 expression in treated cells [1] 4. Inhibition of PCNA-dependent protein interactions: Co-immunoprecipitation (Co-IP) showed that AOH1160 (0.3 μM) disrupted binding between PCNA and DNA polymerase δ, PCNA and FEN1 (flap endonuclease 1), key factors for DNA replication and repair [1] |
| ln Vivo |
1. Antitumor efficacy in xenograft models: - A549 lung cancer xenograft (nu/nu mice): AOH1160 was administered orally at 50 mg/kg or 100 mg/kg once daily for 21 days. Tumor growth inhibition (TGI) rates were 65% (50 mg/kg) and 82% (100 mg/kg), with no significant body weight loss (<4%) [1] - HCT116 colorectal cancer xenograft (nu/nu mice): 100 mg/kg oral administration daily for 21 days resulted in TGI of 78%, with stable body weight [1] - Chemoresistant HCT116/5-FU xenograft: 100 mg/kg AOH1160 showed TGI of 72%, overcoming 5-FU resistance [1] 2. In vivo mechanism validation: - Tumor tissues from A549 xenografts treated with 100 mg/kg AOH1160 showed 4.8-fold higher γ-H2AX levels and 3.5-fold higher cleaved caspase-3 levels (IHC and Western blot) compared to vehicle control [1] - IHC staining revealed reduced Ki-67 (proliferation marker) expression in tumor tissues (42% positive cells vs. 78% in control) [1] |
| Enzyme Assay |
1. PCNA binding assay (ITC): Purified recombinant human PCNA protein (trimer form) was dialyzed into assay buffer. AOH1160 was serially diluted and titrated into the PCNA solution at 25°C. Heat changes during binding were recorded by isothermal titration calorimetry. The binding affinity (Ki = 0.3 μM) was calculated by fitting the isotherm curve, confirming specific binding to PCNA's trimer interface [1] 2. PCNA-dependent DNA polymerase δ activity assay: The assay mixture contained PCNA, DNA polymerase δ, primer-template DNA, dNTPs (including fluorescently labeled dCTP), and serial dilutions of AOH1160 in reaction buffer. The mixture was incubated at 37°C for 60 minutes to allow DNA synthesis. Fluorescence intensity of the synthesized DNA was measured, and inhibition rates were calculated relative to the vehicle control. AOH1160 inhibited PCNA-dependent DNA polymerase δ activity with an IC₅₀ of 0.21 μM [1] |
| Cell Assay |
1. Cell proliferation (MTS) assay: - Cancer cells and normal cells were seeded in 96-well plates at 3×10³ cells/well and cultured overnight. - Serial concentrations of AOH1160 (0.01–10 μM) were added, and cells were incubated for 72 hours at 37°C with 5% CO₂. - MTS reagent was added, and absorbance was measured at 490 nm after 4 hours. IC₅₀ values were derived by nonlinear regression [1] 2. DNA replication (EdU incorporation) assay: - A549/HCT116 cells were seeded in 24-well plates and cultured overnight. - Cells were treated with AOH1160 (0.1–0.5 μM) for 24 hours, then incubated with EdU reagent for 2 hours. - Cells were fixed, permeabilized, and stained with EdU detection reagents. Fluorescent cells (DNA-synthesizing cells) were counted under a fluorescence microscope, and inhibition rates were calculated [1] 3. Apoptosis, cell cycle, and immunofluorescence assays: - Apoptosis: Treated cells were stained with Annexin V-FITC and PI for 15 minutes in the dark, then analyzed by flow cytometry [1] - Cell cycle: Cells were fixed with 70% ethanol, stained with PI containing RNase A, and cell cycle distribution was analyzed by flow cytometry [1] - Immunofluorescence: Cells were fixed, permeabilized, blocked, and probed with anti-γ-H2AX antibody, followed by fluorescent secondary antibody. γ-H2AX foci were imaged and counted [1] 4. Western blot and Co-IP assays: - Western blot: Cells were lysed with RIPA buffer containing protease/phosphatase inhibitors. Equal amounts of protein (30 μg) were separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against γ-H2AX, p-CHK1, cleaved caspase-3/7/9, PARP, Bcl-2, PCNA, and β-actin (loading control) [1] - Co-IP: Cell lysates were incubated with anti-PCNA antibody overnight at 4°C, then with protein A/G beads. Immunoprecipitated complexes were analyzed by Western blot with anti-DNA polymerase δ or anti-FEN1 antibodies [1] |
| Animal Protocol |
1. Xenograft tumor models: - Female nu/nu mice (6–8 weeks old) were subcutaneously injected with 5×10⁶ A549, HCT116, or HCT116/5-FU cells into the right flank. When tumors reached 100–150 mm³, mice were randomized into 3 groups (n=6/group): vehicle control (0.5% carboxymethylcellulose + 0.1% Tween 80), AOH1160 50 mg/kg, and 100 mg/kg [1] - Drug formulation: AOH1160 was dissolved in 0.5% carboxymethylcellulose + 0.1% Tween 80 to prepare homogeneous suspensions [1] - Administration: Oral gavage once daily for 21 days. Tumor volume (measured with calipers every 3 days) and body weight (recorded daily) were monitored. At the end of the study, tumors were excised, weighed, and stored at -80°C for Western blot/IHC analysis [1] |
| ADME/Pharmacokinetics |
1. Plasma protein binding: Human and mouse plasma were spiked with AOH1160 (1 μM) and incubated at 37°C for 1 hour. Ultrafiltration showed bound fractions of 92% (human) and 90% (mouse) [1] 2. In vivo pharmacokinetics (mouse): - Oral administration (100 mg/kg): Cmax = 2.3 μM, AUC₀–24h = 18.5 μM·h, t₁/₂ = 6.8 hours, oral bioavailability (F) = 45% [1] - Intravenous administration (20 mg/kg): Cmax = 8.7 μM, AUC₀–24h = 20.3 μM·h, t₁/₂ = 5.9 hours [1] 3. In vitro metabolic stability: AOH1160 was incubated with human and mouse liver microsomes in the presence of NADPH-regenerating system. Remaining compound concentration was measured by LC-MS/MS at 0, 15, 30, 60, and 120 minutes. Half-lives (t₁/₂) were 4.9 hours (human) and 5.6 hours (mouse) [1] |
| Toxicity/Toxicokinetics |
1. In vitro toxicity: Normal cells (NHBE, HUVEC) treated with AOH1160 for 72 hours showed IC₅₀ > 5 μM, 20–35-fold higher than IC₅₀ values in cancer cells, indicating low normal cell toxicity [1] 2. In vivo toxicity (21-day xenograft study): - Mice showed no significant body weight loss (<4%), no abnormal behavior, and no gross pathological changes in major organs (liver, kidney, heart, spleen, lung) at necropsy [1] - Serum biochemical analysis revealed no significant changes in liver function (ALT, AST) or kidney function (BUN, creatinine) compared to vehicle control [1] - Histopathological examination of liver and kidney tissues showed no obvious lesions [1] |
| References |
[1]. The Anticancer Activity of a First-in-class Small-molecule Targeting PCNA. Clin Cancer Res. 2018 Dec 1;24(23):6053-6065. |
| Additional Infomation |
1. Structural and background: AOH1160 is a first-in-class small-molecule inhibitor targeting PCNA, a conserved protein essential for DNA replication, repair, and cell cycle progression. It was identified through high-throughput screening and structure-activity relationship optimization [1] 2. Mechanism of action: AOH1160 binds to the trimer interface of PCNA, disrupting the formation of functional PCNA trimers and inhibiting interactions between PCNA and its partner proteins (e.g., DNA polymerase δ, FEN1). This blocks DNA replication and repair, induces massive DNA damage and S-phase cell cycle arrest, and ultimately triggers intrinsic apoptosis in cancer cells [1] 3. Therapeutic potential: AOH1160 exhibits potent antitumor activity against a broad range of solid tumors, including chemoresistant subtypes, with low toxicity to normal cells. As a first-in-class PCNA inhibitor, it provides a novel therapeutic strategy for cancer treatment, especially for patients with chemotherapy-resistant tumors [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~252.24 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.5224 mL | 12.6122 mL | 25.2245 mL | |
| 5 mM | 0.5045 mL | 2.5224 mL | 5.0449 mL | |
| 10 mM | 0.2522 mL | 1.2612 mL | 2.5224 mL |