Physicochemical Properties
| Molecular Formula | C13H8CL2N2 |
| Molecular Weight | 263.122020721436 |
| Exact Mass | 262.006 |
| CAS # | 931417-26-4 |
| PubChem CID | 11565176 |
| Appearance | Light yellow to yellow solid powder |
| LogP | 3.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 17 |
| Complexity | 345 |
| Defined Atom Stereocenter Count | 0 |
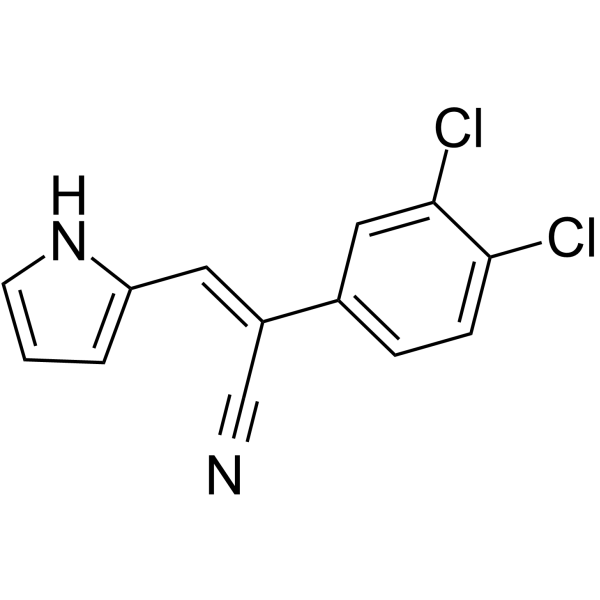
| SMILES | C(/C1C=CC(Cl)=C(Cl)C=1)(\C#N)=C\C1NC=CC=1 |
| InChi Key | IJJHHDLGGYDXGD-UXBLZVDNSA-N |
| InChi Code | InChI=1S/C13H8Cl2N2/c14-12-4-3-9(7-13(12)15)10(8-16)6-11-2-1-5-17-11/h1-7,17H/b10-6+ |
| Chemical Name | (Z)-2-(3,4-dichlorophenyl)-3-(1H-pyrrol-2-yl)prop-2-enenitrile |
| Synonyms | ANI7; ANI 7 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In MCF10A and MDA-MB-468 cells, ANI-7 (2.5 μM; 24 h) treatment significantly arrested the S-phase and G2 + M-phase cell cycle with minimal effects. in MCF10A cells from normal breasts [1]. DNA double-strand damage was indicated by a large increase in H2AXɣ and a considerable rise in CHK2 level and phosphorylation in MDA-MB-468 cells after treatment with ANI-7 (2 μM) for 12-24 hours [1]. Enhancing the effects of ANI-7 is AhR pathway inhibition. AhR and CYP1 member expression, as well as XRE activity, are activated by ANI-7 [1]. A comparison of GI50 values revealed that MCF-7 cells produced a GI50 value of 0.38 μM in response to ANI-7, while lung, colon, ovarian, neuronal, glial, prostate, and pancreatic cell lines showed values of 3.0. -42 micrometers. The A431 vulvar cell line (GI50 of 0.51 μM) is the only other tumor type that exhibits notable growth inhibition by ANI-7 [1][1]. ANI-7 essentially failed to inhibit the growth of MDA-MB-231 and MCF10A cells (GI50 range 17-26μM), but it moderately inhibits the growth of BT20 and BT474 cells (GI50 range 1-2μM), and effectively inhibits the growth of T47D, ZR-75-1, MCF-7, SKBR3, and MDA-MB-468 breast cancer cells (GI50 range 0.16-0.38μM). Furthermore, drug-resistant cells (MCF-7/VP16: GI50 of 0.21 μM) can still not be grown by ANI-7 [1]. |
| Cell Assay |
Cell cycle analysis[1] Cell Types: MCF10A and MDA-MB-468 Cell Tested Concentrations: 2.5 μM Incubation Duration: 24 hrs (hours) Experimental Results: Induction of significant S phase and G2 + M phase cell cycle arrest in MDA-MB-468 cells , the effect on normal breast MCF10A cells is negligible. Western Blot Analysis [1] Cell Types: MDA-MB-468 Cell Tested Concentrations: 2 μM Incubation Duration: 12 hrs (hours), 24 hrs (hours) Experimental Results: resulted in a significant increase in the content and phosphorylation of CHK2 (25-fold increase), and induced H2AXɣ Dramatically increased (by 3.5 times). |
| References |
[1]. (Z)-2-(3,4-Dichlorophenyl)-3-(1H-Pyrrol-2-yl)Acrylonitrile Exhibits Selective Antitumor Activity in Breast Cancer Cell Lines via the Aryl Hydrocarbon Receptor Pathway. Mol Pharmacol. 2018 Feb;93(2):168-177. [2]. Dichlorophenylacrylonitriles as AhR Ligands That Display Selective Breast Cancer Cytotoxicity in vitro. ChemMedChem. 2018 Jul 18;13(14):1447-1458. [3]. Library synthesis and cytotoxicity of a family of 2-phenylacrylonitriles and discovery of an estrogen dependent breast cancer lead compound. Medicinal Chemistry Communication. January 20112. (1):31-37. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~20.83 mg/mL (~79.17 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.91 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (7.91 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.8005 mL | 19.0027 mL | 38.0055 mL | |
| 5 mM | 0.7601 mL | 3.8005 mL | 7.6011 mL | |
| 10 mM | 0.3801 mL | 1.9003 mL | 3.8005 mL |