AMG9810 (AMG-9810) is a nove, potent, selective and competitive antagonist of vanilloid receptor 1 (TRPV1) with cancer-promoting effects. It inhibits TRPV1 with IC50 values of 24.5 and 85.6 nM for human and rat TRPV1, repectively. AMG9810 promotes mouse skin tumor development. The topical application of AMG9810 resulted in a significant increase in the expression level of the epidermal growth factor receptor (EGFR) and its downstream Akt/mammalian target of rapamycin (mTOR)-signaling pathway. This increase was not only observed in AMG9810-treated tumor tissue but was also found in skin tissue treated with AMG9810. In telomerase-immortalized primary human keratinocytes, AMG9810 promoted proliferation that was mediated through the EGFR/Akt/mTOR-signaling pathway. In summary, our data suggest that the TRPV1 antagonist, AMG9810, promotes mouse skin tumorigenesis mediated through EGFR/Akt/mTOR signaling. Thus, the application of this compound for pain relief might increase the risk of skin cancer.
Physicochemical Properties
| Molecular Formula | C21H23NO3 |
| Molecular Weight | 337.4122 |
| Exact Mass | 337.167 |
| CAS # | 545395-94-6 |
| PubChem CID | 680502 |
| Appearance | White to light yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 512.5±50.0 °C at 760 mmHg |
| Flash Point | 263.8±30.1 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.618 |
| LogP | 5.47 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 25 |
| Complexity | 474 |
| Defined Atom Stereocenter Count | 0 |
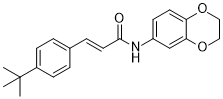
| SMILES | CC(C)(C)C1=CC=C(C=C1)/C=C/C(=O)NC2=CC3=C(C=C2)OCCO3 |
| InChi Key | GZTFUVZVLYUPRG-IZZDOVSWSA-N |
| InChi Code | InChI=1S/C21H23NO3/c1-21(2,3)16-7-4-15(5-8-16)6-11-20(23)22-17-9-10-18-19(14-17)25-13-12-24-18/h4-11,14H,12-13H2,1-3H3,(H,22,23)/b11-6+ |
| Chemical Name | 3-(4-t-Butylphenyl)-N-(2,3-dihydrobenzo(b)(1,4)dioxin-6-yl)acrylamide |
| Synonyms | AMG9810; AMG-9810; AMG 9810. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
IC50: 24.5 nM (human TRPV1), 85.6 nM (rat TRPV1)[1] Transient Receptor Potential Vanilloid 1 (TRPV1) (IC50 = 17 nM in calcium flux assay; Ki = 21 nM in radioligand binding assay) [1] Transient Receptor Potential Vanilloid 1 (TRPV1) [2] |
| ln Vitro |
In cultures of rat dorsal root ganglion primary neurons, AMG9810 blocks capsaicin-evoked depolarization and calcitonin gene-related peptide release. It also inhibits capsaicin-, proton-, heat-, and endogenous ligand-induced uptake of 45Ca2+ into TRPV1-expressing cells. AMG9810 is a competitive antagonist of capsaicin activation (IC50 value for human TRPV1, 24.5±15.7 nM; rat TRPV1, 85.6±39.4 nM). It blocks all known modes of TRPV1 activation, including protons (IC50 value for rat TRPV1, 294±192 nM). AMG 9810 acts as a potent and selective competitive antagonist of TRPV1, effectively inhibiting capsaicin (1 μM)-induced calcium influx in HEK293 cells stably expressing human TRPV1 with an IC50 of 17 nM; it also blocks resiniferatoxin (RTX)- and proton (pH 5.0)-induced TRPV1 activation in the same cell model, with IC50 values of 23 nM and 31 nM, respectively, and shows no significant activity against other TRP channels (TRPV2, TRPV3, TRPA1) at concentrations up to 1 μM [1] AMG 9810 promotes the proliferation of mouse epidermal JB6 Cl41 cells in a dose-dependent manner (1-10 μM), increases colony formation efficiency of JB6 Cl41 cells in soft agar assays (colony number increased by 2.3-fold at 5 μM compared with control), and upregulates the phosphorylation of EGFR (Tyr1068) and Akt (Ser473) detected by Western blotting; this proliferative effect is abrogated by EGFR inhibitors (erlotinib) or Akt inhibitors (LY294002) [2] |
| ln Vivo |
In a model of inflammatory pain caused by intraplantar injection of complete Freund's adjuvant, AMG9810 reverses thermal and mechanical hyperalgesia and is effective in reducing capsaicin-induced eye wiping in a dose-dependent manner. AMG9810 has no discernible impact on motor function at effective levels. In an animal model of inflammatory pain, AMG9810 is the first cinnamide TRPV1 antagonist that has been shown to inhibit capsaicin-induced eye wiping behavior and reverse hyperalgesia[1]. AMG9810 stimulates the growth of cutaneous tumors in mice. The epidermal growth factor receptor (EGFR) and its downstream Akt/mammalian target of rapamycin (mTOR)-signaling pathway are significantly upregulated upon topical administration of AMG9810[2]. In rat models of formalin-induced inflammatory hyperalgesia, intraperitoneal administration of AMG 9810 (1-10 mg/kg) dose-dependently reduces the nociceptive response in both phase 1 (0-5 min) and phase 2 (15-30 min) of the formalin test, with an ED50 of 3.2 mg/kg for phase 2 inhibition; in the chronic constriction injury (CCI) model of neuropathic pain, oral administration of AMG 9810 (3-30 mg/kg) reverses thermal hyperalgesia and mechanical allodynia starting at 1 h post-dosing, with a duration of action exceeding 6 h [1] In the DMBA/TPA-induced mouse skin tumorigenesis model, topical application of AMG 9810 (0.1-1 nmol per mouse, twice weekly) for 20 weeks significantly increases the number of papillomas per mouse (average 12.6 papillomas/mouse at 1 nmol dose vs. 4.2 in vehicle group) and the size of tumors (average diameter 3.8 mm vs. 1.5 mm in vehicle group); immunohistochemical analysis of tumor tissues shows increased phosphorylation of EGFR and Akt in the AMG 9810-treated group, and the tumor-promoting effect is blocked by co-administration of the EGFR inhibitor gefitinib [2] |
| Enzyme Assay |
For radioligand binding assay of TRPV1: Prepare membrane homogenates from HEK293 cells stably expressing human TRPV1, adjust protein concentration to 50 μg/mL in binding buffer; incubate the membrane suspension with [³H]resiniferatoxin (0.5 nM) and serial dilutions of AMG 9810 (10⁻¹²-10⁻⁶ M) at 25°C for 90 min; terminate the reaction by rapid vacuum filtration through glass fiber filters, wash filters three times with cold binding buffer; quantify radioactivity using liquid scintillation counting, calculate Ki values via Cheng-Prusoff equation [1] For calcium flux assay of TRPV1 activity: Seed HEK293-TRPV1 cells in 96-well plates at a density of 1×10⁴ cells/well, culture for 24 h; load cells with calcium-sensitive fluorescent dye for 30 min at 37°C; pre-treat cells with AMG 9810 (10⁻¹¹-10⁻⁶ M) for 10 min, then add capsaicin (1 μM) to trigger calcium influx; measure fluorescence intensity at excitation 485 nm/emission 520 nm using a microplate reader, calculate IC50 values for inhibition of capsaicin-induced fluorescence changes [1] |
| Cell Assay |
To assess cyotoxicity of AMG9810, N/TERT1 cells are treated with different concentrations of AMG9810 (0.25, 0.5, 1, 5 μM) and cultured for various periods of time (24, 48, 72 h). The CellTiter 96 AQueous One Solution is added to each well and then cells are kept in a 37°C, 5% CO2 incubator for 1 h. Absorbance is then measured at 492 and 690 nm with a plate reader[2]. Cultivate HEK293 cells stably expressing human TRPV1 in standard culture medium, seed into 96-well black-walled plates at a density of 1×10⁴ cells per well and incubate for 24 hours to allow cell adhesion; load the cells with a calcium indicator dye for 30 minutes at 37°C in the dark, then pre-incubate with different concentrations of AMG 9810 (ranging from 10 pM to 1 μM) for 10 minutes at room temperature; add capsaicin, resiniferatoxin or acidic buffer (pH 5.0) to the wells to activate TRPV1, and immediately measure the fluorescence intensity of the cells over a 60-second period using a fluorescence microplate reader to assess calcium influx; for specificity testing, repeat the assay using HEK293 cells expressing TRPV2, TRPV3 or TRPA1, and treat with AMG 9810 at concentrations up to 1 μM [1] Culture mouse epidermal JB6 Cl41 cells in complete medium, seed into 96-well plates at a density of 5×10³ cells per well and treat with AMG 9810 at concentrations of 0.1, 1, 5 and 10 μM for 24, 48 and 72 hours; assess cell proliferation using a colorimetric cell viability assay, measure absorbance at 490 nm and calculate the relative proliferation rate compared to vehicle-treated controls; for colony formation assays, seed 1×10³ JB6 Cl41 cells per well in 6-well plates with soft agar, treat with AMG 9810 (1-10 μM) and incubate for 14 days, then count visible colonies under a microscope; for Western blot analysis, treat JB6 Cl41 cells with AMG 9810 (5 μM) for 0, 15, 30, 60 minutes, extract total cellular protein, separate proteins by SDS-PAGE, transfer to membranes and probe with antibodies against phosphorylated EGFR (Tyr1068), total EGFR, phosphorylated Akt (Ser473), total Akt and GAPDH (loading control) [2] |
| Animal Protocol |
For the rat formalin-induced hyperalgesia model: Use male Sprague-Dawley rats (150-200 g), acclimate to the laboratory environment for 7 days before experiments; administer AMG 9810 via intraperitoneal injection at doses of 1, 3, 10 mg/kg (dissolved in a vehicle of 10% DMSO and 90% saline) 30 minutes prior to subcutaneous injection of 5% formalin (50 μL) into the hind paw; record the time the rat spends licking or biting the injected paw during phase 1 (0-5 min) and phase 2 (15-30 min) after formalin administration, and calculate the percentage inhibition of nociceptive behavior compared to vehicle-treated rats [1] For the rat chronic constriction injury (CCI) neuropathic pain model: Induce CCI by ligating the sciatic nerve of male Sprague-Dawley rats (200-250 g) with chromic gut sutures; after 7 days (when hyperalgesia is established), administer AMG 9810 via oral gavage at doses of 3, 10, 30 mg/kg (formulated in 0.5% methylcellulose) once daily for 7 days; measure thermal hyperalgesia using a hot plate test (52°C) and mechanical allodynia using von Frey filaments at 1, 3, 6, 24 hours post-first dosing [1] For the mouse skin tumorigenesis model: Use female CD-1 mice (6-8 weeks old), initiate tumor formation by a single topical application of 7,12-dimethylbenz[a]anthracene (DMBA, 50 μg in 100 μL acetone) to the shaved dorsal skin; one week later, start twice-weekly topical applications of 12-O-tetradecanoylphorbol-13-acetate (TPA, 10 μg in 100 μL acetone) to promote tumor growth, and co-administer AMG 9810 topically at doses of 0.1, 0.5, 1 nmol per mouse (dissolved in 100 μL acetone) immediately after each TPA application for 20 weeks; monitor the number and size of papillomas weekly, and at the end of the experiment, harvest tumor tissues for immunohistochemical analysis of phosphorylated EGFR and Akt [2] |
| References |
[1]. AMG9810 [(E)-3-(4-t-butylphenyl)-N-(2,3-dihydrobenzo[b][1,4] dioxin-6-yl)acrylamide] a novel vanilloid receptor 1 (TRPV1) antagonist with antihyperalgesic properties. J Pharmacol Exp Ther. 2005 Apr;313(1):474-84. [2]. TRPV1-antagonist AMG9810 promotes mouse skin tumorigenesis through EGFR/Akt signaling. Carcinogenesis. 2011 May;32(5):779-85. |
| Additional Infomation |
3-(4-tert-butylphenyl)-N-(2,3-dihydro-1,4-benzodioxin-6-yl)-2-propenamide is a member of cinnamamides and a secondary carboxamide. AMG 9810 is a novel, potent and selective competitive antagonist of the vanilloid receptor 1 (TRPV1), with a chemical structure of (E)-3-(4-t-butylphenyl)-N-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)acrylamide; it was developed as a potential therapeutic agent for the treatment of chronic pain due to its antihyperalgesic properties in preclinical pain models [1] The antihyperalgesic mechanism of AMG 9810 is based on the selective blockade of TRPV1, a cation channel expressed in sensory neurons that mediates nociceptive signaling in response to heat, acid and capsaicin [1] AMG 9810 was found to promote skin tumorigenesis in mice via activation of the EGFR/Akt signaling pathway, suggesting that long-term use of TRPV1 antagonists may carry a potential risk of skin cancer development, which limits its clinical application for chronic pain treatment [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 33 mg/mL (~97.80 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.41 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.41 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.41 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9638 mL | 14.8188 mL | 29.6375 mL | |
| 5 mM | 0.5928 mL | 2.9638 mL | 5.9275 mL | |
| 10 mM | 0.2964 mL | 1.4819 mL | 2.9638 mL |