AMG-337 is a novel, potent and highly selective small molecule ATP-competitive MET kinase inhibitor. In enzymatic assays, AMG 337 inhibited MET kinase activity with an IC50 of < 5nM nM. MET kinase activity was inhibited by AMG 337, with an IC50 of less than 5 nM nM. In a competitive binding assay, AMG 337 was profiled against a broad panel of over 400 protein and lipid kinases and showed remarkable selectivity for MET. AMG 337 inhibited HGF-dependent MET phosphorylation in cellular assays, showing an IC50 of less than 10 nM. AMG 337 was profiled in cell viability assays with a diverse panel of more than 200 cancer cell lines in order to find predictive genomic markers of response. Only two stomach cancer cell lines (SNU-5 and Hs746T), both of which have MET gene amplification, were impacted by treatment with AMG 337 in terms of viability. In all tested cell lines, the AMG 337 IC50 was > 10 μM, while it was < 50 nM in the two sensitive cell lines. In MET-amplified gastric cancer xenograft models, oral administration of AMG 337 produced a strong dose-dependent anti-tumor efficacy in vivo. This tumor growth inhibition was consistent with the pharmacodynamic modulation of MET signaling. To sum up, these results highlight the potential therapeutic use of AMG 337 as a treatment for tumors exhibiting dysregulated MET signaling, including MET amplification. AMG 337 is presently being studied in patients with solid tumors for safety, tolerability, and pharmacokinetics in a phase 1 clinical trial.
Physicochemical Properties
| Molecular Formula | C23H22FN7O3 |
| Molecular Weight | 463.4643 |
| Exact Mass | 463.176 |
| Elemental Analysis | C, 59.60; H, 4.78; F, 4.10; N, 21.16; O, 10.36 |
| CAS # | 1173699-31-4 |
| Related CAS # | 1173699-31-4 |
| PubChem CID | 44181686 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.689 |
| LogP | 1.42 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 34 |
| Complexity | 760 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | FC1=C([H])C(C2C([H])=NN(C([H])([H])[H])C=2[H])=C([H])N2C1=NN=C2[C@@]([H])(C([H])([H])[H])N1C([H])=C([H])C2=C(C([H])=C(C([H])=N2)OC([H])([H])C([H])([H])OC([H])([H])[H])C1=O |
| InChi Key | DWHXUGDWKAIASB-CQSZACIVSA-N |
| InChi Code | InChI=1S/C23H22FN7O3/c1-14(30-5-4-20-18(23(30)32)9-17(11-25-20)34-7-6-33-3)21-27-28-22-19(24)8-15(13-31(21)22)16-10-26-29(2)12-16/h4-5,8-14H,6-7H2,1-3H3/t14-/m1/s1 |
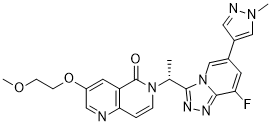
| Chemical Name | 6-[(1R)-1-[8-fluoro-6-(1-methylpyrazol-4-yl)-[1,2,4]triazolo[4,3-a]pyridin-3-yl]ethyl]-3-(2-methoxyethoxy)-1,6-naphthyridin-5-one |
| Synonyms | AMG337; AMG 337; AMG-337 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
MET receptor (IC50 = 1 nM); MET(H1094R) (IC50 = 1 nM); MET(M1250T) (IC50 = 4.7 nM); MET(V1092I) (IC50 = 21.5 nM)
AMG 337 targets MET (mesenchymal-epithelial transition factor) receptor tyrosine kinase (biochemical IC₅₀ = 1.2 nM for human MET kinase activity [1] ; cellular IC₅₀ = 2-10 nM for MET phosphorylation inhibition in MET-amplified cancer cells [1][2] ; >400-fold selectivity over other kinases in kinase profiling [1] |
| ln Vitro |
AMG 337 inhibits WT MET and a subset of MET mutants present in papillary renal cell carcinoma with a potent effect on their enzymatic activity. It is most likely due to a disruption of the inactive confirmation of the activation loop in the MET kinase domain that AMG 337 is unable to inhibit the Y1230 and D1228 mutants. Moreover, AMG 337 has an IC50 of 5 nM against PC3 cells, which inhibits HGF-induced MET phosphorylation in cells. In cancer cell lines that rely on MET, AMG 337 prevents cell division. In MET-amplified gastric cancer cell lines, AMG 337 inhibits signaling via the PI3K and MAPK pathways, which has a significant impact on cell survival and proliferation[1]. 1. AMG 337 potently inhibited MET kinase activity in biochemical assays with an IC₅₀ of 1.2 nM, showing >400-fold selectivity for MET over other kinases tested [1] 2. In MET-dependent cancer cell lines (MKN45, SNU-5, SNU-620, MHCC97H, HCCLM3), AMG 337 (10-100 nM) completely inhibited MET phosphorylation within 2 hours of treatment, leading to dose-dependent inhibition of downstream signaling (Gab-1, ERK1/2, AKT) [1][2] 3. Antiproliferative activity in MET-amplified cancer cells: - Gastric cancer: SNU-5 (IC₅₀ = 2 nM), SNU-620 (IC₅₀ = 5 nM), MKN45 (IC₅₀ = 10 nM) [1] - Hepatocellular carcinoma: MHCC97H (IC₅₀ = 15 nM), HCCLM3 (IC₅₀ = 25 nM) [2] - No significant inhibition in non-MET-amplified cells (IC₅₀ > 10 μM) [1][2] 4. Cell cycle analysis in SNU-620 cells showed AMG 337 (100 nM) induced G₁ phase arrest with decreased S phase DNA synthesis after 24 hours [1] 5. Apoptosis induction in sensitive cells: AMG 337 (100 nM) increased cleaved PARP and caspase-3 levels after 24 hours in SNU-5 and SNU-620 cells, but not in MKN45 cells [1] 6. In HCCLM3 hepatocellular carcinoma cells, AMG 337 (10-100 nM) inhibited cell migration and invasion in transwell assays by 60-80% [2] |
| ln Vivo |
AMG 337 demonstrates remarkable effectiveness, as it inhibits Gab-1 phosphorylation by over 90% at a dosage of 0.75 mg/kg, or 32 nmol/L of free drug concentration. It is possible that AMG 337 possesses the preclinical qualities necessary to investigate the function of MET in human cancer because it is well tolerated when given at doses that correspond with total MET inhibition for a full day[1]. 1. In TPR-MET mouse xenograft model (constitutively active MET): - AMG 337 (0.5-10 mg/kg PO daily) caused dose-dependent inhibition of tumor growth - 0.75 mg/kg dose achieved >90% inhibition of Gab-1 phosphorylation in tumors [1] 2. In MET-amplified gastric cancer xenografts: - SNU-5 model: AMG 337 (0.3 mg/kg daily) produced 100% growth inhibition; 1-10 mg/kg caused tumor regression [1] - U-87 MG glioblastoma model: 3 mg/kg daily inhibited growth by 90%, 10 mg/kg caused complete regression [1] 3. In hepatocellular carcinoma PDX models: - LI0612 (MET-amplified): AMG 337 (3-30 mg/kg daily) inhibited tumor growth with TGI of 101-120%, with 10-30 mg/kg causing tumor regression [2] - LI1078 (non-MET-amplified): no significant growth inhibition at any dose tested (TGI < 17%) [2] 4. Pharmacodynamic studies in tumors showed AMG 337 (3-10 mg/kg) inhibited MET and Gab-1 phosphorylation by >80% within 3 hours of dosing, with effects lasting up to 24 hours [1][2] |
| Enzyme Assay |
1. MET kinase activity assay: Recombinant human MET kinase domain was incubated with ATP (10 μM), biotinylated substrate peptide, and AMG 337 (0.01-100 nM) in kinase buffer at 30°C for 60 minutes; reactions were terminated with EDTA, and phosphorylated peptides were captured on streptavidin plates and detected by phospho-specific antibody and chemiluminescence; IC₅₀ values were calculated from dose-response curves [1] 2. Kinase selectivity profiling: AMG 337 (100 nM) was tested against >400 different kinases using a similar assay format; selectivity was determined by comparing IC₅₀ values for MET versus other kinases [1] 3. AlphaScreen assay for MET pathway inhibition: Cell lysates from AMG 337-treated cells were incubated with AlphaScreen beads coated with antibodies against p-MET or p-Gab-1; signal intensity was measured on a multimode reader to quantify pathway inhibition [2] |
| Cell Assay |
Cells are seeded in 96-well plates at an ideal density to guarantee proliferation throughout the course of the experiments in order to assess the impact of AMG 337 on viability. AMG 337 is diluted 10-fold, three times, serially, and applied to cells for 72 hours at a maximum concentration of 3 mmol/L. The CellTiter-Glo Luminescent Cell Viability Assay is used to quantify viability. 1. Cell viability/proliferation assay: Cancer cells (5×10³/well) were seeded in 96-well plates and treated with AMG 337 (0.01-10 μM) for 72 hours; viability was measured by MTT reduction or ATP content; IC₅₀ values were calculated for each cell line [1][2] 2. Western blot analysis for MET pathway inhibition: Cells were treated with AMG 337 (10-100 nM) for 2-24 hours; whole cell lysates were prepared, separated by SDS-PAGE, and probed with antibodies against p-MET, total MET, p-Gab-1, p-ERK, p-AKT, and loading controls (actin); band intensities were quantified by densitometry to determine inhibition of signaling [1][2] 3. Cell cycle analysis: SNU-620 cells were treated with AMG 337 (100 nM) for 24 hours, fixed with ethanol, stained with propidium iodide, and analyzed by flow cytometry to determine distribution of cells in G₁, S, and G₂/M phases [1] 4. Apoptosis detection: Cells were treated with AMG 337 (100 nM) for 24 hours, stained with Annexin V-FITC and propidium iodide, and analyzed by flow cytometry to quantify apoptotic cells; western blot was also performed to detect cleaved PARP and caspase-3 [1] 5. Migration/invasion assay: HCCLM3 cells were treated with AMG 337 (10-100 nM) for 24 hours, then seeded in transwell chambers with Matrigel (for invasion) or without (for migration); after 24 hours, cells on the lower surface were fixed, stained, and counted [2] |
| Animal Protocol |
Female CD1 nu/nu or athymic nude mice(Tumor xenograft models) 0.1, 0.5, 0.75, 1, 2, or 3 mg/kg by oral gavage 1. Xenograft tumor model: - Mice (6-8 weeks old) were implanted subcutaneously with 1×10⁶ cancer cells (SNU-5, SNU-620, U-87 MG) or patient-derived tumor fragments (LI0612, LI1078) - When tumors reached 100-200 mm³, mice were randomized into treatment groups - AMG 337 was formulated in 30% hydroxypropyl-β-cyclodextrin + 10% Pluronic F68 and administered orally by gavage at 0.3-30 mg/kg daily for 14 days - Tumor volume was measured twice weekly (volume = length × width² × 0.5) - At study end, tumors were harvested for western blot analysis of p-MET and p-Gab-1 [1][2] 2. Pharmacodynamic study: - Mice bearing established xenografts were treated with a single dose of AMG 337 (3-10 mg/kg) - Tumors were harvested at 3, 6, 12, and 24 hours post-dosing - Protein lysates were prepared for western blot analysis to determine duration of MET pathway inhibition [1][2] |
| ADME/Pharmacokinetics |
1. In mice: - Oral bioavailability: 85% - Terminal half-life: 4.2 hours - Volume of distribution: 1.8 L/kg - Plasma clearance: 0.3 L/h/kg [1] 2. In rats: - Cmax = 450 nM (10 mg/kg PO) - Tmax = 1 hour - AUC = 1200 nM·h - Oral bioavailability = 78% - Tumor/plasma concentration ratio = 0.8 at 1 hour post-dosing [1][2] 3. Metabolism: AMG 337 was primarily metabolized by CYP3A4 (70%) and CYP2D6 (20%) in human liver microsomes; metabolites showed <10% activity against MET [1] 4. Plasma protein binding: >95% in human plasma [1] 5. Excretion: <5% of the dose was excreted unchanged in urine; >90% was excreted as metabolites in feces [1] |
| Toxicity/Toxicokinetics |
1. Acute toxicity: No mortality or significant adverse effects were observed in mice at single oral doses up to 2000 mg/kg or intravenous doses up to 100 mg/kg [1] 2. Subchronic toxicity: - Rats treated with AMG 337 (30-100 mg/kg/day PO for 28 days) showed no significant changes in body weight, food intake, or clinical chemistry parameters (ALT, AST, BUN, creatinine) - No treatment-related histopathological changes were observed in major organs (liver, kidney, heart, lung, spleen) [1] 3. Cardiac safety: AMG 337 (1-10 μM) showed no significant inhibition of hERG potassium channel in patch clamp assays, indicating low risk for QT prolongation [1] 4. Genotoxicity: Negative results in Ames test, chromosome aberration assay, and micronucleus test [1] |
| References |
[1]. In Vitro and In Vivo Activity of AMG 337, a Potent and Selective MET Kinase Inhibitor, in MET-Dependent Cancer Models. Mol Cancer Ther. 2016 Jul;15(7):1568-79. [2]. Preclinical Evaluation of AMG 337, a Highly Selective Small Molecule MET Inhibitor, in Hepatocellular Carcinoma. Mol Cancer Ther. 2016 Jun;15(6):1227-37. |
| Additional Infomation |
AMG-337 is under investigation in clinical trial NCT03147976 (QUILT-3.036: AMG 337 in Subjects With Advanced or Metastatic Solid Tumors). c-Met Inhibitor AMG 337 is an orally bioavailable inhibitor of the proto-oncogene c-Met with potential antineoplastic activity. c-Met inhibitor AMG 337 selectively binds to c-Met, thereby disrupting c-Met signal transduction pathways. This may induce cell death in tumor cells overexpressing c-Met protein or expressing constitutively activated c-Met protein. c-Met protein, the product of the proto-oncogene c-Met, is a receptor tyrosine kinase also known as hepatocyte growth factor receptor (HGFR); this protein is overexpressed or mutated in many tumor cell types and plays key roles in tumor cell proliferation, survival, invasion, and metastasis, and tumor angiogenesis. 1. AMG 337 is a novel, oral, ATP-competitive MET kinase inhibitor designed for the treatment of MET-driven cancers [1][2] 2. Mechanism of action: Binds to the ATP-binding pocket of MET kinase, preventing phosphorylation of MET and downstream signaling (Gab-1, PI3K/AKT, MAPK/ERK), leading to G₁ cell cycle arrest and apoptosis in sensitive cells [1] 3. Biomarker strategy: MET amplification (FISH ratio >2.0) and high MET protein expression (IHC 3+) are predictive biomarkers for response to AMG 337 [2] 4. Clinical development: Entered Phase I clinical trials in patients with advanced solid tumors, showing promising efficacy in MET-amplified gastric and esophageal cancers [1][2] 5. Therapeutic potential: Being investigated for treatment of non-small cell lung cancer, hepatocellular carcinoma, and other MET-driven malignancies [1][2] |
Solubility Data
| Solubility (In Vitro) |
DMSO: 95~100 mg/mL (~205.0 mM) Ethanol: ~95 mg/mL (~205.0 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.39 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1577 mL | 10.7884 mL | 21.5768 mL | |
| 5 mM | 0.4315 mL | 2.1577 mL | 4.3154 mL | |
| 10 mM | 0.2158 mL | 1.0788 mL | 2.1577 mL |