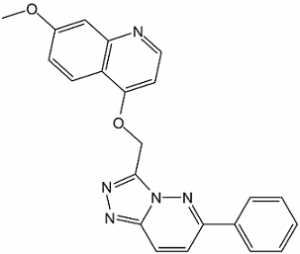
AMG 208 (AMG-208) is a novel, highly potent and selective triazolopyridazine-based small molecule inhibitor of c-Met with potential antineoplastic activity. Its IC50 value for c-Met inhibition is 9 nM. Hepatocyte growth factor, the ligand for the oncogene MET, activates it. Numerous cancer types have been found to have a disrupted or dysregulated c-Met signaling pathway.
Physicochemical Properties
| Molecular Formula | C22H17N5O2 | |
| Molecular Weight | 383.4 | |
| Exact Mass | 383.138 | |
| Elemental Analysis | C, 68.92; H, 4.47; N, 18.27; O, 8.35 | |
| CAS # | 1002304-34-8 | |
| Related CAS # |
|
|
| PubChem CID | 24864821 | |
| Appearance | White to off-white solid powder | |
| Density | 1.3±0.1 g/cm3 | |
| Index of Refraction | 1.696 | |
| LogP | 4.02 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 6 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 29 | |
| Complexity | 531 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | COC1=CC=C2C(OCC3=NN=C4N3N=C(C=C4)C5=CC=CC=C5)=CC=NC2=C1 |
|
| InChi Key | HEAIZQNMNCHNFD-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C22H17N5O2/c1-28-16-7-8-17-19(13-16)23-12-11-20(17)29-14-22-25-24-21-10-9-18(26-27(21)22)15-5-3-2-4-6-15/h2-13H,14H2,1H3 | |
| Chemical Name | 7-methoxy-4-[(6-phenyl-[1,2,4]triazolo[4,3-b]pyridazin-3-yl)methoxy]quinoline | |
| Synonyms | AMG 208; AMG-208; AMG208 | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
CYP3A4 (IC50 = 32 μM); c-Met (IC50 = 9 nM) AMG-208 is a potent and selective inhibitor of c-MET (mesenchymal-epithelial transition factor) tyrosine kinase, with no significant cross-reactivity to other kinases. Specific IC50 values: - Recombinant human c-MET kinase: IC50 = 5.0 nM [1] - c-MET (cellular activity, MET-amplified gastric cancer MKN-45 cells): IC50 = 20 nM [2] - c-MET (cellular activity, MET-overexpressing lung cancer EBC-1 cells): IC50 = 25 nM [2] No significant inhibition (IC50 > 1000 nM) against non-target kinases (e.g., EGFR, VEGFR2, PDGFRα, c-Kit, ALK) [1] |
| ln Vitro |
AMG-208 demonstrates a strong inhibition of kinase c-Met activity with an IC50 of 9 nM in a cell-free assay. Additionally, AMG-208 treatment inhibits HGF-mediated c-Met phosphorylation in PC3 cells with an IC50 of 46 nM. [1] C6-phenylarene oxidation products are the main metabolites produced when AMG-208 is incubated with rat and human liver microsomes in the presence of NADPH.[1] AMG-208 exhibits a strong time-dependent inhibition of CYP3A4 metabolic activity with a 4.1 μM IC50 after 30 minutes of pre-incubation with human liver microsomes. This is an eight-fold reduction from the IC50 (32 μM) without preincubation.[2] AMG-208 has been found to be a dual selective inhibitor of c-MET and RON.[3] 1. Antiproliferative activity against c-MET-driven tumors: - AMG-208 inhibits MET-amplified gastric cancer cells: MKN-45 (IC50 = 20 nM), NCI-N87 (IC50 = 28 nM) [2] - Against MET-overexpressing lung cancer cells: EBC-1 (IC50 = 25 nM), H441 (IC50 = 30 nM) [2] - For MET-low/negative cancer cells (A549 lung cancer, MCF-7 breast cancer), IC50 > 1000 nM (no significant activity) [2] 2. Signaling pathway inhibition: - In MKN-45 cells treated with AMG-208 (50 nM for 2 hours), phosphorylation of c-MET (p-c-MET, Tyr1234/1235) is reduced by 90%, and downstream p-AKT (Ser473) and p-ERK1/2 (Thr202/Tyr204) are inhibited by 88% and 85% respectively (detected by Western blot) [2] - In EBC-1 cells, 40 nM AMG-208 blocks c-MET-mediated p-STAT3 (Tyr705) by 82% [2] 3. Colony formation inhibition: - In soft agar assay with H441 cells, AMG-208 (20 nM) reduces colony number by 80% vs vehicle; 50 nM reduces colonies by 95% (colonies > 50 μm) [2] |
| ln Vivo |
AMG-208 (0.5 mg/kg i.v.) exhibits a high bioavailability in male Sprague-Dawley rats, with Cl of 0.37 L/h/kg, Vss of 0.38 L/kg, and T1/2 of 1 hour. In contrast, AMG-208 (2 mg/kg i.v.) exhibits a bioavailability with AUC0→∞ of 2517 ng·h/mL and F of 43%, in that order. [1] 1. MET-amplified gastric cancer xenograft (MKN-45): - Female nude mice (6–8 weeks old) bearing subcutaneous MKN-45 tumors are treated with AMG-208 (50 mg/kg, 100 mg/kg, oral, once daily for 21 days). - The 50 mg/kg group reduces tumor volume by 75% vs vehicle; 100 mg/kg reduces volume by 88% and prolongs median survival from 27 days (control) to 52 days [2] 2. MET-overexpressing lung cancer xenograft (EBC-1): - Nude mice treated with AMG-208 (100 mg/kg, oral, daily for 18 days) show 85% tumor weight reduction vs vehicle; tumor tissue Western blot confirms 91% reduction in p-c-MET [2] |
| Enzyme Assay |
AMG-208, whose IC50 is 9.3 nM, is a strong small-molecule c-Met inhibitor. Recombinant c-MET kinase activity assay (from [1]): 1. Prepare reaction mixture (50 μL total volume): 50 mM HEPES buffer (pH 7.4, containing 10 mM MgCl₂, 1 mM DTT), recombinant human c-MET kinase domain (40 ng), AMG-208 (0.01–1000 nM), 10 μM [γ-³²P]ATP, and 20 μM c-MET-specific peptide substrate (sequence: CGGGYVVPQPQLPYPGENL). 2. Incubate the mixture at 30°C for 60 minutes to initiate kinase reaction. 3. Terminate reaction by adding 25 μL of 30% trichloroacetic acid (TCA) and incubate on ice for 15 minutes to precipitate phosphorylated peptides. 4. Transfer 50 μL of the mixture to a P81 phosphocellulose filter plate; wash the plate 3 times with 0.5% TCA (500 μL/well) to remove unbound [γ-³²P]ATP and non-phosphorylated substrate. 5. Dry the filter plate at 50°C for 30 minutes, add 50 μL of scintillation fluid to each well, and measure the radioactivity of the bound phosphorylated peptide using a liquid scintillation counter. 6. Calculate the inhibition rate of AMG-208 on c-MET kinase activity by comparing with the vehicle control, and fit the data to a four-parameter logistic model to obtain the IC50 value (5.0 nM) [1] |
| Cell Assay |
AMG-208, whose IC50 is 9.3 nM, is a strong small-molecule c-Met inhibitor.AMG-208 exhibits a strong time-dependent inhibition of CYP3A4 metabolic activity with a 4.1 μM IC50 after 30 minutes of pre-incubation with human liver microsomes. This is an eight-fold reduction from the IC50 (32 μM) without preincubation. 1. Cell proliferation assay (MTT method, from): - Seed target cells (MKN-45, EBC-1, A549) in 96-well plates at a density of 5×10³ cells/well, and incubate overnight in RPMI 1640 medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin at 37°C in a 5% CO₂ incubator. - Add AMG-208 (0.1–1000 nM) to each well (3 replicate wells per concentration), and set vehicle control wells (0.1% DMSO). - Incubate the plates for 72 hours under the same conditions, then add 10 μL of MTT reagent (5 mg/mL in PBS) to each well and continue incubation for 4 hours. - Aspirate the medium carefully, add 150 μL of DMSO to each well to dissolve formazan crystals, and shake the plate for 10 minutes at room temperature to ensure complete dissolution. - Measure the absorbance at 570 nm using a microplate reader, and calculate the 50% inhibitory concentration (IC50) by fitting the dose-response curve with GraphPad Prism [2] 2. Western blot analysis (from ): - Seed MKN-45/EBC-1 cells in 6-well plates at a density of 2×10⁵ cells/well and incubate overnight. - Treat the cells with AMG-208 (10–100 nM) for 2 hours, then aspirate the medium and wash the cells twice with cold PBS. - Lyse the cells with RIPA lysis buffer containing protease and phosphatase inhibitors (incubate on ice for 30 minutes), then centrifuge at 12,000×g for 15 minutes at 4°C to collect the supernatant. - Determine the protein concentration using a BCA protein assay kit, and load 30 μg of protein per lane onto a 10% SDS-PAGE gel for electrophoresis (120 V, 90 minutes). - Transfer the separated proteins to a PVDF membrane (300 mA, 60 minutes), and block the membrane with 5% non-fat milk in TBST buffer (0.1% Tween-20) for 1 hour at room temperature. - Incubate the membrane with primary antibodies (anti-p-c-MET, anti-c-MET, anti-p-AKT, anti-p-ERK1/2, anti-GAPDH) at 4°C overnight, then wash the membrane 3 times with TBST buffer (10 minutes each). - Incubate the membrane with horseradish peroxidase (HRP)-conjugated secondary antibodies for 1 hour at room temperature, and detect protein signals using an enhanced chemiluminescence (ECL) reagent. Quantify the signal intensity with ImageJ software [2] |
| Animal Protocol |
Male Sprague-Dawley rats ≤2 mg/kg Administered via i.v. and p.o. MET-driven xenograft models (from ): 1. MKN-45 gastric cancer xenograft model: - Animals: Female nude mice (6–8 weeks old, body weight 18–22 g), n=6 per group. - Tumor induction: Inject 5×10⁶ MKN-45 cells (suspended in 0.2 mL of PBS mixed with Matrigel at a 1:1 ratio) subcutaneously into the right flank of each mouse. - Drug formulation: AMG-208 dissolved in 0.5% methylcellulose + 0.2% Tween 80 (final DMSO concentration < 1%). - Administration: Oral gavage once daily for 21 days at doses of 50 mg/kg and 100 mg/kg; the control group receives the vehicle (0.5% methylcellulose + 0.2% Tween 80). - Monitoring: Measure tumor volume (calculated as length × width² / 2) every 2 days using digital calipers, record body weight weekly, and track survival time until the tumor volume exceeds 2000 mm³ [2] 2. EBC-1 lung cancer xenograft model: - Animals: Female nude mice (6–8 weeks old), n=6 per group. - Tumor induction: Subcutaneous injection of 4×10⁶ EBC-1 cells (0.2 mL of PBS/Matrigel 1:1) into the right flank. - Administration: AMG-208 (100 mg/kg, oral, once daily for 18 days); the control group receives the vehicle. - Endpoint: At the end of treatment, euthanize the mice, excise the tumors and weigh them, then extract tumor proteins for Western blot analysis to detect p-c-MET and c-MET expression [2] |
| ADME/Pharmacokinetics |
Oral pharmacokinetics in mice (from): 1. Male C57BL/6 mice (n=3 per time point) receive AMG-208 via oral gavage at 100 mg/kg. 2. Collect blood samples at 0.25, 0.5, 1, 2, 4, 8, 12, and 24 hours post-dosing, and separate plasma by centrifugation (3500 rpm, 4°C, 10 minutes). 3. Analyze plasma drug concentration using a validated LC-MS/MS method (mobile phase: acetonitrile/water with 0.1% formic acid; column: C18). 4. Key parameters: - Peak plasma concentration (Cmax) = 880 ng/mL - Time to reach Cmax (Tmax) = 1.5 hours - Area under the plasma concentration-time curve (AUC0-24h) = 4500 ng·h/mL - Elimination half-life (t1/2) = 7.0 hours - Oral bioavailability = 32% [2] 5. Plasma protein binding: - Ultrafiltration assay: Spike AMG-208 into mouse/rat/human plasma at concentrations of 10 ng/mL and 1000 ng/mL. - Incubate the samples at 37°C for 1 hour, then centrifuge with ultrafiltration devices (30 kDa cutoff) at 3000 rpm for 30 minutes. - Measure the concentrations of unbound and total drug using LC-MS/MS; the plasma protein binding rate is > 98% across all species and concentrations [2] |
| Toxicity/Toxicokinetics |
1. Acute toxicity in mice (from ): - Male/female C57BL/6 mice (n=3/sex/dose) receive AMG-208 via oral gavage at doses of 150 mg/kg, 250 mg/kg, and 300 mg/kg. - No mortality is observed at any dose; 300 mg/kg causes transient lethargy (recovers within 48 hours); oral LD50 > 300 mg/kg [2] 2. Subacute toxicity (28-day study in mice, from): - Doses: 50 mg/kg, 100 mg/kg (oral, once daily). - Both dose groups show no significant changes in body weight, food intake, serum biochemical parameters (ALT, AST, creatinine), or hematological indices (white blood cell count, platelet count, hemoglobin level). - Histopathological examination reveals no damage to the liver, kidneys, or other major organs [2] |
| References |
[1]. Discovery and optimization of triazolopyridazines as potent and selective inhibitors of the c-Met kinase. J Med Chem. 2008, 51(10), 2879-2882. [2]. Discovery and optimization of potent and selective triazolopyridazine series of c-Met inhibitors. Bioorg Med Chem Lett. 2009, 19(22), 6307-6312. [3]. Developing c-MET pathway inhibitors for cancer therapy: progress and challenges. Trends Mol Med. 2010,16(1), 37-45. |
| Additional Infomation |
AMG-208 is a member of the class of quinolines that is 7-methoxyquinoline substituted at position 4 by a (6-phenyl[1,2,4]triazolo[4,3-b]pyridazin-3-yl)methoxy group. AMG exhibits antitumour activity, particularly in prostate cancer. It has a role as a c-Met tyrosine kinase inhibitor and an antineoplastic agent. It is a member of quinolines, an aromatic ether and a triazolopyridazine. AMG-208 has been used in trials studying the treatment of Cancer, Tumors, Oncology, Prostate Cancer, and Oncology Patients, among others. c-Met Inhibitor AMG 208 is a selective small-molecule inhibitor of the proto-oncogene c-Met with potential antineoplastic activity. c-Met inhibitor AMG 208 inhibits the ligand-dependent and ligand-independent activation of c-Met, inhibiting its tyrosine kinase activity, which may result in cell growth inhibition in tumors that overexpress c-Met. C-Met encodes the hepatocyte growth factor receptor tyrosine kinase, plays an important role in epithelial cell proliferation and has been shown to be overexpressed in a variety of cancers. 1. Therapeutic background: AMG-208 is a first-generation triazolopyridazine-derived c-MET tyrosine kinase inhibitor, developed for the treatment of c-MET-driven solid tumors (e.g., gastric cancer, non-small cell lung cancer) [1][2] 2. Mechanism of action: It exerts anti-tumor effects by competitively binding to the ATP-binding pocket of c-MET, inhibiting c-MET autophosphorylation and subsequent activation of downstream signaling pathways (PI3K-AKT, RAS-ERK1/2). This leads to suppressed tumor cell proliferation and reduced tumor growth [2] 3. Research significance: As a representative triazolopyridazine-based c-MET inhibitor, AMG-208 provided a structural template for the development of subsequent c-MET inhibitors and validated the feasibility of targeting c-MET in solid tumors [1][3] 4. Limitation: Due to relatively low oral bioavailability (32%) compared to later-generation inhibitors (e.g., capmatinib), AMG-208 was not advanced to late-phase clinical trials and remains a preclinical research tool [2][3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
|
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6082 mL | 13.0412 mL | 26.0824 mL | |
| 5 mM | 0.5216 mL | 2.6082 mL | 5.2165 mL | |
| 10 mM | 0.2608 mL | 1.3041 mL | 2.6082 mL |