Physicochemical Properties
| Molecular Formula | C17H17NO3 |
| Molecular Weight | 283.32 |
| Exact Mass | 283.121 |
| CAS # | 404009-46-7 |
| PubChem CID | 16760391 |
| Appearance | White to yellow solid powder |
| LogP | 2.524 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 21 |
| Complexity | 348 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | FALILNHGILFDLC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C17H17NO3/c19-16-12-14(18-8-10-21-11-9-18)6-7-15(16)17(20)13-4-2-1-3-5-13/h1-7,12,19H,8-11H2 |
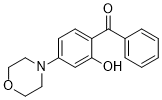
| Chemical Name | (2-hydroxy-4-morpholin-4-ylphenyl)-phenylmethanone |
| Synonyms | AMA-37 AMA37 AMA 37 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Inhibiting PI3K is less successful with AMA-37 [2]. The ability of UCN-01, isogranimide, and debromohymenaldolase to bypass G2 block (ρ < 0.05) is reduced by AMA-37 (20 μM), but not by caffeine [3]. Radiosensitization is the outcome of using AMA37 to inhibit DNA-PK [3]. |
| References |
[1]. A pharmacological map of the PI3-K family defines a role for p110alpha in insulin signaling. Cell. 2006 May 19;125(4):733-47. [2]. Isoform-specific phosphoinositide 3-kinase inhibitors from an arylmorpholine scaffold. Bioorg Med Chem. 2004 Sep 1;12(17):4749-59. [3]. Effect of combined DNA repair inhibition and G2 checkpoint inhibition on cell cycle progression after DNA damage. Mol Cancer Ther. 2006 Apr;5(4):885-92. |
| Additional Infomation | [2-hydroxy-4-(4-morpholinyl)phenyl]-phenylmethanone is a member of benzophenones. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~352.96 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.82 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.82 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5296 mL | 17.6479 mL | 35.2958 mL | |
| 5 mM | 0.7059 mL | 3.5296 mL | 7.0592 mL | |
| 10 mM | 0.3530 mL | 1.7648 mL | 3.5296 mL |