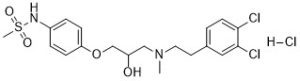
AM-92016 HCl, the hydrochloride salt of AM-92016 (an oxypropanolamine analog of sotalol), is a novel, potent and specific IK inhibitor. It has bpotential Class III anti-arrhythmic properties in-vitro at concentrations showing 1000 times more potency than sotalol. AM 92016 exhibited proarrhythmic rather than antiarrhythmic activity when administered in-vivo to either guinea-pigs or pigs.AM-92016 (hydrochloride) delays rectifier potassium channel (IK), repolarizes the membrane thereby restricting the duration of the nerve impulse. AM 92016 (hydrochloride) prolongs cardiac action potential duration in guinea-pig and rabbit isolated ventricular cells with an IC50 of 30 nM.
Physicochemical Properties
| Molecular Formula | C19H25CL3N2O4S |
| Molecular Weight | 483.8368 |
| Exact Mass | 482.06 |
| CAS # | 133229-11-5 |
| Related CAS # | 133229-23-9;133229-11-5 (HCl); |
| PubChem CID | 45073415 |
| Appearance | Light yellow to yellow solid powder |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 29 |
| Complexity | 553 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | ClC1=C(C([H])=C([H])C(=C1[H])C([H])([H])C([H])([H])N(C([H])([H])[H])C([H])([H])C([H])(C([H])([H])OC1C([H])=C([H])C(=C([H])C=1[H])N([H])S(C([H])([H])[H])(=O)=O)O[H])Cl.Cl[H] |
| InChi Key | TXOARFPCQOBODS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H24Cl2N2O4S.ClH/c1-23(10-9-14-3-8-18(20)19(21)11-14)12-16(24)13-27-17-6-4-15(5-7-17)22-28(2,25)26;/h3-8,11,16,22,24H,9-10,12-13H2,1-2H3;1H |
| Chemical Name | N-(4-(3-((3,4-dichlorophenethyl)(methyl)amino)-2-hydroxypropoxy)phenyl)methanesulfonamide hydrochloride |
| Synonyms | AM92016 HCl; AM-92016 hydrochloride; AM-92016 HCl; AM 92016 HCl; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Time-dependent delayed rectifier potassium current (IK) in cardiac ventricular myocytes (IC50 ≈ 30 nM for IK blockade) [2] |
| ln Vitro |
In single guinea-pig and rabbit ventricular myocytes, compound II (10 nM – 1 µM) concentration-dependently and reversibly prolonged the action potential duration at 20% (APD20) and 90% (APD90) repolarization levels without affecting the resting membrane potential. In guinea-pig cells, it also increased action potential amplitude. The maximum achievable prolongation was 30-35%. [2] Under voltage-clamp conditions, compound II (1 µM) selectively and potently blocked the time-dependent delayed rectifier potassium current (IK), measured as the outward tail current upon repolarization. It did not affect L-type calcium currents (tested in the presence of nisoldipine or using voltage steps), inward rectifier potassium current (IK1) (assessed via ramp voltage protocols), or fast sodium currents (effects preserved in the presence of tetrodotoxin). [2] The log dose-response curves for action potential prolongation and IK blockade by compound II were similar, with an IC50 of approximately 30 nM for IK blockade. Submaximal concentrations (50 nM) did not alter the apparent reversal potential of IK (~ -75 mV), the time constants of IK deactivation, or the kinetics of its time-dependent activation. A small shift (~10 mV depolarizing) in the voltage-dependence of IK activation was observed. [2] |
| Cell Assay |
Ventricular Myocyte Isolation: Single ventricular myocytes were isolated from guinea-pig and rabbit hearts. The hearts were excised and cells were isolated using enzymatic digestion methods. [2] Action Potential Recording: Isolated cells were superfused with oxygenated physiological solution at 36°C. Action potentials were stimulated by 2 ms current pulses at 1 Hz using a single-microelectrode system in bridge mode. Measurements were taken after drug effects reached a steady state (approximately 4 minutes). [2] Voltage-Clamp Recording: A switch-clamp method was used. To isolate IK, cells were held at -50 mV and depolarizing steps were applied to activate the current. IK was measured as the decaying outward tail current upon repolarization. To minimize contaminating calcium-activated currents, experiments were conducted in the presence of the L-type calcium channel blocker nisoldipine (2-3 µM) or by including the calcium chelator BAPTA (75 mM) in the recording electrode. [2] Current-Voltage (I-V) Relationship: The effect on IK1 was assessed by ramping the membrane voltage from -90 mV to +20 mV at a rate of 20 mV/s and recording the resulting current. [2] Kinetic Analysis: The time-dependent activation of IK was studied by applying depolarizing pulses of varying durations. The deactivation kinetics of IK tail currents were analyzed by fitting the decay to a biexponential function. [2] |
| References |
[1]. ConnorsSP,etal.Actions and mechanisms of action of novel analogues of sotalol on guinea-pig and rabbitventricular cells. Br J Pharmacol. 1992 Aug;106(4):958-65. |
| Additional Infomation |
Compound II (1-(4-methanesulphonamidophenoxy)-3-(N-methyl 3,4 dichlorophenylethylamino)-2-propanol) is a prototype of a novel series of sotalol analogues synthesized as potential Class III antiarrhythmic agents. It is devoid of β1-adrenoceptor blocking activity. The primary mechanism for its action potential prolongation effect is potent and selective blockade of the time-dependent delayed rectifier potassium current (IK). It was among the most potent compounds of its class described at the time of publication. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~260 mg/mL (~537.37 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.17 mg/mL (4.48 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.17 mg/mL (4.48 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.17 mg/mL (4.48 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 21.7 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0668 mL | 10.3340 mL | 20.6680 mL | |
| 5 mM | 0.4134 mL | 2.0668 mL | 4.1336 mL | |
| 10 mM | 0.2067 mL | 1.0334 mL | 2.0668 mL |