Physicochemical Properties
| Molecular Formula | C20H21B2NO5 |
| Molecular Weight | 377.006445646286 |
| Exact Mass | 377.16 |
| CAS # | 856849-35-9 |
| PubChem CID | 11639443 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 695.4±65.0 °C at 760 mmHg |
| Flash Point | 374.3±34.3 °C |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.644 |
| LogP | 4.14 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 28 |
| Complexity | 556 |
| Defined Atom Stereocenter Count | 0 |
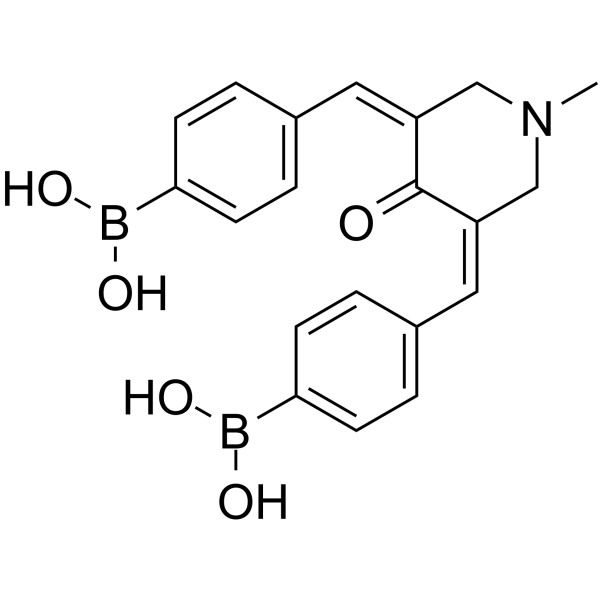
| SMILES | B(O)(O)C1=CC=C(C=C1)/C=C\2/C(=O)/C(=C/C3=CC=C(C=C3)B(O)O)/CN(C2)C |
| InChi Key | SRPIKXGUPAKTIZ-OTYYAQKOSA-N |
| InChi Code | InChI=1S/C20H21B2NO5/c1-23-12-16(10-14-2-6-18(7-3-14)21(25)26)20(24)17(13-23)11-15-4-8-19(9-5-15)22(27)28/h2-11,25-28H,12-13H2,1H3/b16-10+,17-11+ |
| Chemical Name | [4-[(E)-[(5E)-5-[(4-boronophenyl)methylidene]-1-methyl-4-oxopiperidin-3-ylidene]methyl]phenyl]boronic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | At a dose of 1 µM, treatment with proteasome inhibitor IX (AM114; 0.1-10 µM; 14 days; HCT116 p53+/+ cells) led to a reduction in cell viability in p53+/+ and p53-/- cells of about 70% and 20%, respectively [1]. In colony formation experiments, proteasome inhibitor IX (AM114) demonstrated strong activity against p53+/+ cells, with an IC50 value of 0.6 µM [1]. The proteasome inhibitor IX (1 µM) was incubated with HCT116 p53+/+ cells (AM114). After 48 hours, 28% of the cells had positive Annexin V staining, and after 72 hours, 76% of the cells were dead[1]. Proteasome inhibitor IX (AM114) treatment prevents the 20S proteasome from acting in a chymotrypsin-like manner in vitro, which causes ubiquitinated p53 and other cellular proteins to accumulate significantly inside the cell [1]. |
| References |
[1]. A Boronic-Chalcone Derivative Exhibits Potent Anticancer Activity Through Inhibition of the Proteasome. Mol Pharmacol. 2006 Jul;70(1):426-33. [2]. Green Tea Polyphenols Block the Anticancer Effects of Bortezomib and Other Boronic Acid-Based Proteasome Inhibitors. Blood. 2009 Jun 4;113(23):5927-37. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~6.25 mg/mL (~16.58 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.62 mg/mL (1.64 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 6.2 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 0.62 mg/mL (1.64 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 6.2 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6524 mL | 13.2622 mL | 26.5245 mL | |
| 5 mM | 0.5305 mL | 2.6524 mL | 5.3049 mL | |
| 10 mM | 0.2652 mL | 1.3262 mL | 2.6524 mL |