Physicochemical Properties
| Molecular Formula | C10H13CLFN3O4 |
| Molecular Weight | 293.6793 |
| Exact Mass | 293.057 |
| CAS # | 1445379-92-9 |
| Related CAS # | Lumicitabine;1445385-02-3 |
| PubChem CID | 71621663 |
| Appearance | White to off-white solid powder |
| Density | 1.8±0.1 g/cm3 |
| Boiling Point | 532.8±60.0 °C at 760 mmHg |
| Flash Point | 276.0±32.9 °C |
| Vapour Pressure | 0.0±3.2 mmHg at 25°C |
| Index of Refraction | 1.668 |
| LogP | -0.42 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 19 |
| Complexity | 447 |
| Defined Atom Stereocenter Count | 4 |
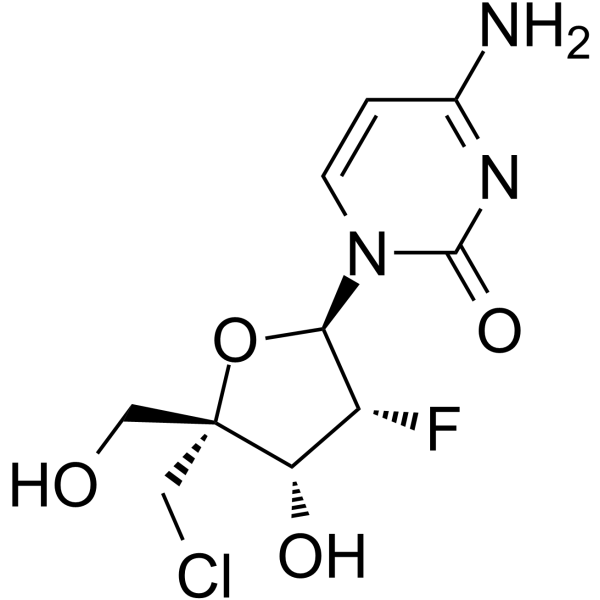
| SMILES | C1=CN(C(=O)N=C1N)[C@H]2[C@@H]([C@@H]([C@](O2)(CO)CCl)O)F |
| InChi Key | AWSRKKBIPSQHOJ-IBCQBUCCSA-N |
| InChi Code | InChI=1S/C10H13ClFN3O4/c11-3-10(4-16)7(17)6(12)8(19-10)15-2-1-5(13)14-9(15)18/h1-2,6-8,16-17H,3-4H2,(H2,13,14,18)/t6-,7+,8-,10-/m1/s1 |
| Chemical Name | 4-amino-1-[(2R,3R,4R,5R)-5-(chloromethyl)-3-fluoro-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]pyrimidin-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The drug's active form, ALS-8112 -TP, disables RSV polymerase by causing RNA synthesis to terminate at the 5'-triphosphate [2]. After entering different kinds of respiratory tract epithelial cells, ALS-008112 is phosphorylated to produce intracellular nucleoside triphosphates, which have a half-life of roughly 29. RSV replication is similarly inhibited by hourly nucleoside triphosphates via chain termination [3]. With an IC50 of 0.020 ± 0.008 μM, ALS-8112 is an external RSV replicating ubiquitin ALS-8112-TP that proportionally suppresses the RSV–RNP complex's RNA regulatory action [4]. |
| References |
[1]. Discovery of 4'-chloromethyl-2'-deoxy-3',5'-di-O-isobutyryl-2'-fluorocytidine (ALS-8176), a first-in-class RSV polymerase inhibitor for treatment of human respiratory syncytial virus infection. J Med Chem. 2015 Feb 26;58(4):1862-78. [2]. Activation Pathway of a Nucleoside Analog Inhibiting Respiratory Syncytial Virus Polymerase. ACS Chem Biol. 2017 Jan 20;12(1):83-91. [3]. Activity of Oral ALS-008176 in a Respiratory Syncytial Virus Challenge Study. N Engl J Med. 2015 Nov 19;373(21):2048-58. [4]. Molecular Basis for the Selective Inhibition of Respiratory Syncytial Virus RNA Polymerase by 2'-Fluoro-4'-Chloromethyl-Cytidine Triphosphate. PLoS Pathog. 2015 Jun 22;11(6):e1004995. |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 47 mg/mL (~160.04 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.51 mM) (saturation unknown) in 5% DMSO + 40% PEG300 + 5% Tween80 + 50% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.51 mM) (saturation unknown) in 5% DMSO + 95% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4051 mL | 17.0253 mL | 34.0507 mL | |
| 5 mM | 0.6810 mL | 3.4051 mL | 6.8101 mL | |
| 10 mM | 0.3405 mL | 1.7025 mL | 3.4051 mL |