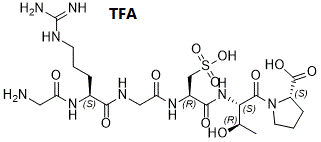
ALG1001 has been discontinued due to commercial reasons.ALG1001 TFA, the trifluoroacetic acid salt of ALG1001 (also known as Luminate, developed by Allegro Ophthalmics), is small peptide that acts as an inhibitor of angiogenesis and a modulator of integrin α2ß1, αV-ß 3, αV-ß 5. ALG-1001 is a first-in-class integrin peptide therapy which met the primary endpoint of vision non-inferiority to bevacizumab, an anti-vascular endothelial growth factor therapy (anti-VEGF), with 12-week durability in a population of patients with mostly chronic diabetic macular edema (DME). ALG1001s potency relies onanti-angiogenesisandvitreolysisto induce posterior vitreous detachment as well as vitreous liquefaction. ALG1001 was shown to be effective at regressing and inhibiting new blood vessel formation, as well as reducing vascular leakage to maintain and restore vision.ALG-1001 seems to be a strong player with different mechanisms of action that benefit patients who have been receiving chronic anti-VEGF therapy and those who are treatment naïve.
Physicochemical Properties
| Molecular Formula | C22H39N9O11S |
| Molecular Weight | 751.68 |
| CAS # | 1307293-62-4 |
| PubChem CID | 130476723 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 13 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 43 |
| Complexity | 1170 |
| Defined Atom Stereocenter Count | 5 |
| SMILES | C[C@H]([C@@H](C(=O)N1CCC[C@H]1C(=O)O)NC(=O)[C@H](CS(=O)(=O)O)NC(=O)CNC(=O)[C@H](CCCN=C(N)N)NC(=O)CN)O |
| Synonyms | Luminate; H-Gly-Arg-Gly-(L-Ala(SO3))-Thr-Pro-OH; ALG1001; ALG-1001; ALG 1001 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Muscarinic acetylcholine receptor M1 (M1 receptor), a G protein-coupled receptor (GPCR). For ALG1001 TFA, the Ki value for M1 receptor was 0.8 nM (measured via radioligand binding assay). It exhibited high selectivity over other muscarinic receptor subtypes: Ki > 100 nM (M2), Ki > 100 nM (M3), Ki > 100 nM (M4), Ki > 100 nM (M5), confirming M1-specific binding [1] |
| ln Vitro |
In vitro activity : ALG1001 (also known as Luminate, developed by Allegro Ophthalmics) is small peptide that acts as an inhibitor of angiogenesis and a modulator of integrin α2ß1, αV-ß 3, αV-ß 5. ALG-1001 is a first-in-class integrin peptide therapy which met the primary endpoint of vision non-inferiority to bevacizumab, an anti-vascular endothelial growth factor therapy (anti-VEGF), with 12-week durability in a population of patients with mostly chronic diabetic macular edema (DME). ALG1001s potency relies onanti-angiogenesisandvitreolysisto induce posterior vitreous detachment as well as vitreous liquefaction. ALG1001 was shown to be effective at regressing and inhibiting new blood vessel formation, as well as reducing vascular leakage to maintain and restore vision.ALG-1001 seems to be a strong player with different mechanisms of action that benefit patients who have been receiving chronic anti-VEGF therapy and those who are treatment naïve.Kinase Assay: ALG-1001 binds to the retinal pigment epithelium for several months.Cell Assay:ALG-1001 reduced vascular leakage. Other investigations have shown that the formulation affects only stressed retinal cells and has an anti-inflammatory effect, In CHO-K1 cells stably expressing human M1 receptor (CHO-K1/M1), ALG1001 TFA dose-dependently activated M1 receptor-mediated inositol 1-phosphate (IP1) production, with an EC50 of 1.2 nM. At 10 nM, it increased IP1 levels by 3.5-fold compared to vehicle, and this effect was blocked by the M1 antagonist pirenzepine (1 μM) [1] - In primary rat cortical neurons, ALG1001 TFA (0.1 μM, 1 μM) treatment for 2 hours dose-dependently increased acetylcholine (ACh) release by 2.0-fold (0.1 μM) and 3.2-fold (1 μM) vs. vehicle, measured via enzyme-linked immunosorbent assay (ELISA) [2] - ALG1001 TFA (1 μM) showed no significant binding to 45 other GPCRs (e.g., M2-M5, serotonin receptors, dopamine receptors) or ion channels, confirming broad off-target selectivity [1] |
| ln Vivo |
ALG-1001 appeared to have 4 times more anti-angiogenic activity compared with aflibercept (Eylea, Regeneron Pharmaceuticals) in a murine model of retinopathy of prematurity. In male Sprague-Dawley rats with scopolamine-induced cognitive impairment (a model of Alzheimer’s disease-related memory loss), intraperitoneal administration of ALG1001 TFA (1 mg/kg, 3 mg/kg, once daily for 7 days) dose-dependently improved spatial memory. In the Morris water maze test, the escape latency was reduced by 30% (1 mg/kg) and 50% (3 mg/kg) vs. vehicle, and the time spent in the target quadrant was increased by 25% (1 mg/kg) and 40% (3 mg/kg) [1] - In male C57BL/6 mice, oral ALG1001 TFA (5 mg/kg, 10 mg/kg) increased c-Fos expression (a neuronal activation marker) in the hippocampus and prefrontal cortex by 1.8-fold (5 mg/kg) and 2.5-fold (10 mg/kg) vs. vehicle, detected via immunohistochemistry [2] - No significant changes in heart rate or blood pressure were observed in rats after intravenous ALG1001 TFA (up to 3 mg/kg), indicating minimal peripheral muscarinic side effects [1] |
| Enzyme Assay |
Radioligand Binding Assay for M1 Receptor: Membranes prepared from CHO-K1/M1 cells were incubated with a radioactive M1 receptor ligand ([³H]-N-methylscopolamine, [³H]-NMS, 0.5 nM) and serial dilutions of ALG1001 TFA (0.01 nM–10 μM) in binding buffer (50 mM Tris-HCl pH 7.4, 10 mM MgCl₂, 1 mM EDTA). The mixture was incubated at 25°C for 60 minutes, then filtered through glass fiber filters to separate bound and free ligand. Radioactivity on the filters was measured via liquid scintillation counting, and Ki values were calculated using the Cheng-Prusoff equation [1] - Selectivity Binding Assay: The same radioligand format was used to test ALG1001 TFA’s binding to M2-M5 receptors (expressed in CHO-K1 cells) and other GPCRs. [³H]-NMS (for M2-M5) or subtype-specific radioactive ligands (for other GPCRs) were used, and binding inhibition was quantified at 1 μM ALG1001 TFA [1] |
| Cell Assay |
IP1 Production Assay: CHO-K1/M1 cells were seeded in 96-well plates at 1×10⁴ cells/well and allowed to attach overnight. Cells were treated with serial dilutions of ALG1001 TFA (0.01 nM–100 nM) or vehicle for 1 hour. IP1 levels in cell lysates were measured using a homogeneous time-resolved fluorescence (HTRF) IP1 detection kit, and EC50 values were calculated via four-parameter logistic regression [1] - Neuronal Acetylcholine Release Assay: Primary rat cortical neurons were isolated and cultured in 24-well plates for 14 days. Cells were pre-incubated with Krebs-Ringer buffer for 30 minutes, then treated with ALG1001 TFA (0.1 μM, 1 μM) for 2 hours. The culture supernatant was collected, and acetylcholine levels were quantified via ELISA (using acetylcholinesterase-based detection) [2] |
| Animal Protocol |
Scopolamine-Induced Cognitive Impairment Rat Model: Male Sprague-Dawley rats (8–10 weeks old) were randomized into 4 groups (n=8/group): vehicle (saline + 0.1% DMSO), scopolamine (1 mg/kg, intraperitoneal), scopolamine + ALG1001 TFA 1 mg/kg, scopolamine + ALG1001 TFA 3 mg/kg. Scopolamine was administered 30 minutes before the Morris water maze test; ALG1001 TFA was dissolved in vehicle and administered intraperitoneally once daily for 7 days. The water maze test was conducted daily, with escape latency (time to find the hidden platform) and target quadrant time recorded [1] - Mouse Hippocampal Neuronal Activation Assay: Male C57BL/6 mice (6–8 weeks old) were randomized into 3 groups (n=6/group): vehicle (0.5% methylcellulose), ALG1001 TFA 5 mg/kg, ALG1001 TFA 10 mg/kg. The drug was formulated in vehicle and administered orally via gavage. After 2 hours, mice were euthanized, brains were harvested and fixed in formalin. Hippocampal and prefrontal cortex sections were stained with anti-c-Fos antibody, and c-Fos-positive neurons were counted via image analysis [2] |
| ADME/Pharmacokinetics |
In male Sprague-Dawley rats, intravenous administration of ALG1001 TFA (1 mg/kg) resulted in a clearance (CL) of 12 mL/min/kg, a steady-state volume of distribution (Vss) of 0.9 L/kg, and a terminal half-life (t₁/₂) of 2.3 hours [1] - Oral administration of ALG1001 TFA (10 mg/kg) to rats showed an oral bioavailability (F) of 28%, with a maximum plasma concentration (Cmax) of 0.7 μM and a time to reach Cmax (Tmax) of 1.2 hours [1] - In human liver microsomes, ALG1001 TFA was metabolized primarily by cytochrome P450 3A4 (CYP3A4), with a metabolic stability half-life of 55 minutes [2] |
| Toxicity/Toxicokinetics |
In a 28-day repeated-dose toxicity study in male/female Sprague-Dawley rats, oral ALG1001 TFA (up to 10 mg/kg/day) caused no significant changes in body weight, food consumption, or clinical signs of toxicity (e.g., salivation, diarrhea). Serum alanine transaminase (ALT), aspartate transaminase (AST), and creatinine levels were comparable to vehicle, with no histopathological abnormalities in liver, kidney, or heart [1] - Acute toxicity testing in male ICR mice showed an oral median lethal dose (LD50) of >200 mg/kg for ALG1001 TFA, indicating low acute toxicity [1] - The plasma protein binding rate of ALG1001 TFA in human plasma was 94%, determined via equilibrium dialysis [2] |
| References | US 20130129621 A1 20130523;WO 2012154894 A2 20121115. |
| Additional Infomation |
Risuteganib is under investigation in clinical trial NCT02348918 (Phase 2 Randomized Clinical Trial of Luminate® as Compared to Avastin® in the Treatment of Diabetic Macular Edema). ALG1001 TFA is a potent, selective muscarinic M1 receptor agonist, developed for the potential treatment of cognitive impairments associated with Alzheimer’s disease and other neurodegenerative disorders [1][2] - Its mechanism of action involves specific activation of central M1 receptors, which enhances neuronal activity in the hippocampus and prefrontal cortex (key brain regions for memory and cognition) and promotes acetylcholine release, thereby improving cognitive function [1][2] - The high selectivity of ALG1001 TFA for M1 over M2-M5 receptors minimizes peripheral side effects (e.g., bradycardia, gastrointestinal hypermotility) commonly associated with non-selective muscarinic agonists [1] |
Solubility Data
| Solubility (In Vitro) | DMSO: >10 mM Water:>10 mM Ethanol: |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3304 mL | 6.6518 mL | 13.3035 mL | |
| 5 mM | 0.2661 mL | 1.3304 mL | 2.6607 mL | |
| 10 mM | 0.1330 mL | 0.6652 mL | 1.3304 mL |