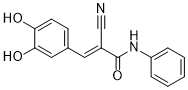
AG 494 sodium is a novel and potent tyrphostin that blocks Cdk2
Physicochemical Properties
| Molecular Formula | C16H12N2O3 |
| Molecular Weight | 280.28 |
| Exact Mass | 280.085 |
| CAS # | 139087-53-9 |
| PubChem CID | 5328771 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.416g/cm3 |
| Boiling Point | 586.9ºC at 760mmHg |
| Flash Point | 308.8ºC |
| LogP | 2.625 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 21 |
| Complexity | 446 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | N#C/C(/C(NC1=CC=CC=C1)=O)=C\C1=CC=C(O)C(O)=C1 |
| InChi Key | HKHOVJYOELRGMV-XYOKQWHBSA-N |
| InChi Code | InChI=1S/C16H12N2O3/c17-10-12(8-11-6-7-14(19)15(20)9-11)16(21)18-13-4-2-1-3-5-13/h1-9,19-20H,(H,18,21)/b12-8+ |
| Chemical Name | (E)-2-cyano-3-(3,4-dihydroxyphenyl)-N-phenylprop-2-enamide |
| Synonyms | AG-494; AG494 sodium; AG 494 Tyrphostin B48Tyrphostin AG-494 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | EGFR tyrosine kinase (IC50=0.7 μM). |
| ln Vitro |
AG 494 suppresses EGF-dependent DNA synthesis and Cdk2 activation in DHER-14 cells [2]. AG-494 both dramatically decreases and prevents NF-kB activation in H2O2-treated cells as well as silica-stimulated cells [4]. ALP activity produced by BMP9 is inhibited in a dose-dependent manner by AG-494 (3-9 μM; 5-7 days) [5]. We have previously shown that the EGFR kinase selective tyrphostin AG494 fails to inhibit EGFR kinase in intact cells. Yet, AG494proved to inhibit EGF- or serum-induced cell proliferation (Osherov et al., J. Biol. Chem. 268 (1993) 11134–11142). In this preliminary communication we show that AG 494 as well as its close analogs AG 490 and AG 555 block Cdk2 activation. In contrast, AG 1478, a more selective EGFR kinase blocker which is also active as EGFR kinase blocker in intact cells, fails to do so. AG 494 exerts its full inhibitory activity on Cdk2 activation even when added 20 h subsequent to EGF addition when Cdk2 activation is maximal. The inhibitory activity on Cdk2 activation parallels its DNA synthesis inhibitory activity, strongly suggesting that its target is one of the molecular mechanisms involved in Cdk2 activation. AG 494 and its analogs may become useful lead compounds for the development of drugs aimed at the cell cycle machinery. [2] BMP9-induced osteogenic differentiation can be effectively blocked by EGFR inhibitors [4] Epidermal growth factor initiates downstream events by activating its tyrosine kinase receptor EGFR (aka HER1). We sought to determine if commonly EGFR inhibitors would exert any effect on BMP9-induced osteogenic differentiation of MSCs. We tested four EGFR inhibitors, AG494, AG1478, Erlotinib and Gefitinib, and found all of them inhibited BMP9-induced ALP activity in a dose-dependent manner (Fig. 3A). Among the four tested inhibitors, Erlotinib and Gefitinib are clinically used as anticancer drugs, which were shown to be slightly more effective than AG494 and AG1478 in terms of inhibiting BMP9-induced ALP activity (Fig. 3A and B). Thus, these in vitro findings suggest that EGFR signalling may play an important role in BMP9-regulated osteogenic differentiation, although these inhibitors may also target other tyrosine kinase. |
| Enzyme Assay |
Inhibition of autophosphorylation [2] Membranes (2 μg/assay) were pre-incubated with EGF (50 nM) in 50 mM HEPES (pH 7.4), 125 mM NaCl, for 15 min at 4°C. The assay was initiated by addition of 8 μl membranes to 12 μl reaction mixture containing 50 mM HEPES (pH 7.4), 125 mM NaCl, 2 μM ATP (=2 K m final), 1 μCi [32P]γ-ATP, 4 mM MnCl2, 24 mM MgCl2, and 4 μl tyrphostin dissolved in 10% Me2SO/45% ethanol/45% DDW (double-distilled water). The assay was conducted at 4°C and terminated after 30 s by the addition of 8 μl boiling SDS-PAGE sample buffer. Proteins were separated on 6% SDS-PAGE, and autoradiography performed. A single EGF inducible 32P-labeled band comigrating in the molecular weight of the receptor could then be detected. This band was scanned and quantified by densitometry. IC50 values were calculated using the E-Z fit program. All reactions were performed rapidly in V-shaped 96-well plates using a multipipette. Reactions were all linear for 30 s. The exposure times of the autoradiograms were controlled so that film exposure was linear. Basal EGF-independent activity did not exceed 5% of the EGF-activated autophosphorylation of HER-1/EGF receptors. Inhibition of pp60c-src activity was essentially performed as described by using the DHER-14 cells as described in detail previously, using denatured enolase as a substrate. Inhibition of autophosphorylation of wheat-germ agglutinin (WGA) purified insulin-receptor was performed as described by Schechter et al. Inhibition of autophosphorylation of HER-2/Neu receptor was performed using membranes prepared from NIH-3T3 cells using published procedures ransfected with the human HER-2/Neu receptor as described for HER-1. Immunoprecipitation and Cdk2 kinase assays [2] DHER-14 cells were grown to confluence for 48 h in Costar 6-well dishes in DMEM +10% calf serum as described. Cells were starved for 3 days. During the last 24 h, cells were activated by various mitogens (20 nM EGF; 50 ng/ml TPA phorbol ester) or 50 μM lysophosphatidic acid (LPA) for various time periods. Cells were incubated with tyrphostins as described for each experiment. Cells were washed once in PBS, then lysed on ice using 1 ml/well cold IP buffer (50 mM HEPES, 250 mM NaCl, 5 mM DTT, 0.25% NP-40, 10 mM NaF, and protease inhibitors). Lysates were incubated on ice for 30 min, then centrifuged 14 000×g for 15 min in a microfuge. Supernatants were transferred, and incubated for 2 h in the presence of a saturating concentration of 1 μg/eppendorf anti-Cdk2 antibodies. The immunocomplex was collected on protein A agarose beads (7.5 μl packed volume per eppendorf) and washed 3 times in IP buffer. The pellet was then washed a fourth time in kinase buffer (50 mM HEPES, 10 mM MgCl, 1 mM DTT, 1 μM cold ATP). All buffer was aspirated from the beads with a fine bore Hamilton syringe, and the kinase reaction initiated by the addition of 40 μl/eppendorf reaction mixture containing kinase buffer and 1 μCi/assay [γ-32P]ATP and 2.5 μg Histone H1 (freshly prepared) per assay. The reaction was performed for 20 min at 30°C, then stopped by addition of 12.5 μl/assay SDS-PAGE sample buffer ×4. Samples were separated on 10% SDS PAGE gels and exposed to film for ≈2 h at −70°C. Quantitation was performed by densitometry. |
| Cell Assay |
Inhibition of [3H]thymidine uptake [2] Cells were seeded at 7000 cells/well in 96-well Costar dishes precoated with 1 μg/well human fibronectin. The cells were grown to confluence for 2 days. The medium was changed to DMEM containing 0.25% calf serum for 48 h and the cells were then incubated 16 h with either 20 nM EGF, 50 μM lysophosphatidic acid or 50 ng/ml TPA (phorbol ester). After 16 h, [3H]thymidine 0.5 μCi/ml was added for 4 h. Different concentrations of tyrphostins dissolved at ×100 concentration in 10% Me2SO were added either 30 min prior to the addition of mitogens, or during the last 4 h along with [3H]thymidine addition. The trichloroacetic acid- precipitable material was quantified by scintillation counting. Basal EGF-independent activity did not exceed 5% of mitogen-dependent [3H]thymidine uptake activity of DHER-14 cells. Tyrphostins were added either 30 min prior to EGF addition or at the time of [3H]thymidine addition. Thus AG494 and AG 1478 were present either for 20 or 4 h. Immunoprecipitation and immunoblot analysis [2] Cells were plated at 2×105 cells/5 ml/well in Costar 6-well dishes, grown to confluence for 2 days and then starved in 2 ml/well DMEM containing 0.25% calf serum for 48 h. Tyrphostins dissolved at ×100 concentration in 10% Me2SO were added during the last 16 h or the last 2 h of starvation. EGF (20 nM) was then added and the plates were incubated for 2 min at 4°C with EGF (20 nM), during which time receptor autophosphorylation is linear. The reaction was terminated by addition of 1 ml/well stop lysis buffer containing 20 mM HEPES (pH 7.4), 125 mM NaCl, 1% Triton X-100, 5 mM NaF, 100 μM NaVO3, 200 μM ZnCl2, 1 mM EDTA, 2 mM EGTA, 1 mM PMSF, 10 μg/ml aprotinin, 5 μg/ml leupeptin. Immunoprecipitation using monoclonal antibody 108 was performed as described above. The immunocomplex was released in boiling sample buffer, and then samples were run on SDS-PAGE 5–15% gradient gels, transferred to nitrocellulose (ABN semi-dry blot), blocked for 30 min in TBST (Tris buffer saline (pH 7.4), 1% Tween-20, 5% bovine serum albumin), then probed with monoclonal anti-phosphotyrosine antibodies PT66 for 3 h at 40°C in TBST, washed 4 times in TBS, and then reproved with [125I]goat anti-mouse antibodies (1 μCi/ml). The blots were then washed 4 times in TBS, air dried and analyzed by autoradiography. |
| References |
[1]. Tyrphostins. 2. Heterocyclic and alpha-substituted benzylidenemalononitrile tyrphostins as potent inhibitors of EGF receptor and ErbB2/neu tyrosine kinases. J Med Chem. 1991;34(6):1896-1907. [2]. Tyrphostin AG 494 blocks Cdk2 activation. FEBS Lett. 1997;410(2-3):187-190. [3]. Selective inhibition of the epidermal growth factor and HER2/neu receptors by tyrphostins. J Biol Chem. 1993 May 25;268(15):11134-42. [4]. Cross-talk between EGF and BMP9 signalling pathways regulates the osteogenic differentiation of mesenchymal stem cells. J Cell Mol Med. 2013;17(9):1160-1172. [5]. SILICA-INDUCED NUCLEAR FACTOR- k B ACTIVATION: INVOLVEMENT OF REACTIVE OXYGEN SPECIES AND PROTEIN TYROSINE KINASE ACTIVATION. Journal of Toxicology and Environmental Health, Part A. Journal of Toxicology and Environmental Health, Part A. Volume 60, 2000 - Issue 1 |
| Additional Infomation |
Tyrphostin B48 is a member of the tyrphostin family of tyrosine kinase inhibitors that inhibits epidermal growth factor receptor. We have previously shown that the EGFR kinase selective tyrphostin AG494 fails to inhibit EGFR kinase in intact cells. Yet, AG 494 proved to inhibit EGF- or serum-induced cell proliferation (Osherov et al., J. Biol. Chem. 268 (1993) 11134-11142). In this preliminary communication we show that AG 494 as well as its close analogs AG 490 and AG 555 block Cdk2 activation. In contrast, AG 1478, a more selective EGFR kinase blocker which is also active as EGFR kinase blocker in intact cells, fails to do so. AG 494 exerts its full inhibitory activity on Cdk2 activation even when added 20 h subsequent to EGF addition when Cdk2 activation is maximal. The inhibitory activity on Cdk2 activation parallels its DNA synthesis inhibitory activity, strongly suggesting that its target is one of the molecular mechanisms involved in Cdk2 activation. AG 494 and its analogs may become useful lead compounds for the development of drugs aimed at the cell cycle machinery. [2] Mesenchymal stem cells (MSCs) are multipotent progenitors, which give rise to several lineages, including bone, cartilage and fat. Epidermal growth factor (EGF) stimulates cell growth, proliferation and differentiation. EGF acts by binding with high affinity to epidermal growth factor receptor (EGFR) on the cell surface and stimulating the intrinsic protein tyrosine kinase activity of its receptor, which initiates a signal transduction cascade causing a variety of biochemical changes within the cell and regulating cell proliferation and differentiation. We have identified BMP9 as one of the most osteogenic BMPs in MSCs. In this study, we investigate if EGF signalling cross-talks with BMP9 and regulates BMP9-induced osteogenic differentiation. We find that EGF potentiates BMP9-induced early and late osteogenic markers of MSCs in vitro, which can be effectively blunted by EGFR inhibitors Gefitinib and Erlotinib or receptor tyrosine kinase inhibitors AG-1478 and AG494 in a dose- and time-dependent manner. Furthermore, EGF significantly augments BMP9-induced bone formation in the cultured mouse foetal limb explants. In vivo stem cell implantation experiment reveals that exogenous expression of EGF in MSCs can effectively potentiate BMP9-induced ectopic bone formation, yielding larger and more mature bone masses. Interestingly, we find that, while EGF can induce BMP9 expression in MSCs, EGFR expression is directly up-regulated by BMP9 through Smad1/5/8 signalling pathway. Thus, the cross-talk between EGF and BMP9 signalling pathways in MSCs may underline their important roles in regulating osteogenic differentiation. Harnessing the synergy between BMP9 and EGF should be beneficial for enhancing osteogenesis in regenerative medicine. [4] Nuclear factor-kB (NF-kB) is a multiprotein complex that may regulate a variety of inflammatory cytokines involved in the initiation and progression of silicosis. The present study documents the ability of in vitro silica exposure to induce DNA-binding activity of NF-kB in a mouse peritoneal macrophage cell line (RAW264.7 cells) and investigates the role of reactive oxygen species (ROS) and/or protein tyrosine kinase in this activation. In vitro exposure of mouse macrophages to silica (100 µg/ml) resulted in a twofold increase in ROS production, measured as the generation of chemiluminescence (CL), and caused activation of NF-kB. Silica-induced CL was inhibited 100% by superoxide dismutase (SOD) and 75% by catalase, while NF-kB activation was inhibited by a variety of antioxidants (catalase, superoxide dismutase, alpha-tocopherol, pyrrolidine dithiocarbamate, or N-acetylcysteine). Further evidence for the involvement of ROS in NF-kB activation is that 1 mM H2O2 enhanced NF-kB/DNA binding and that this activation was inhibited by catalase. Specific inhibitors of protein tyrosine kinase, such as herbimycin A, genistein, and AG494, prevented NF-kB activation in silica-treated cells. Genistein and AG494 also reduced NF-kB activation in H2O2-treated cells. Results con firm that tyrosine phosphorylation of several cellular proteins (approximate molecular mass of 39, 58?70, and 103 kD) was increased in silica-exposed macrophages and that genistein inhibited this silica-induced phosphorylation. In contrast, inhibitors of protein kinase A or C, such as H89, staurosporin, calphostin C, and H7, had no marked inhibitory effect on silica-induced NF-kB activation. The results suggest that ROS may play a role in silica-induced NF-kB activation in macrophages and that phosphorylation events mediated by tyrosine kinase may be involved in this activation.[5] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.5679 mL | 17.8393 mL | 35.6786 mL | |
| 5 mM | 0.7136 mL | 3.5679 mL | 7.1357 mL | |
| 10 mM | 0.3568 mL | 1.7839 mL | 3.5679 mL |