Physicochemical Properties
| Molecular Formula | C10H6F3NO2 |
| Molecular Weight | 229.1554 |
| Exact Mass | 229.035 |
| CAS # | 53518-15-3 |
| PubChem CID | 100641 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 314.6±42.0 °C at 760 mmHg |
| Melting Point | 221-222 °C(lit.) |
| Flash Point | 144.1±27.9 °C |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.562 |
| LogP | 1.64 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 16 |
| Complexity | 337 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JBNOVHJXQSHGRL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C10H6F3NO2/c11-10(12,13)7-4-9(15)16-8-3-5(14)1-2-6(7)8/h1-4H,14H2 |
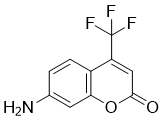
| Chemical Name | 7-amino-4-(trifluoromethyl)chromen-2-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The fluorescent compound N-acetyl-asp-glu-val-asp-7-amino-4-(trifluoromethyl)coumarin (Ac-DEVD-AFC) was used to test caspase activators. The substrate penetrates the cell quickly and is effectively broken down at aspartic acid residues by certain caspases, resulting in the fluorescent molecule 7-amino-4-(trifluoromethyl)coumarin (AFC). The liberated 7-Amino-4-(trifluoromethyl)coumarin was isolated on HPLC and identified by fluorescence upon cell rupture. The intracellular caspase responsible for the emergence of intracellular 7-Amino-4-(trifluoromethyl)coumarin is identified [1] as a burst of the pancaspase benzyloxycarbonyl-val-ala-asp-fluoromethylketone. The synthesis of γ-glutamyl-7-amino-4-(trifluoromethyl)coumarin helped determine the fluorescence of γ-glutamyl transpeptidase by assisting the substrate reaction product 7-amino-4-(trifluoromethyl). With excitation and emission wavelengths of 400 and 490 nm, respectively, and a linear correlation with phosphors in the concentration range of 10-300 pmol/3 mL, coumarin responds linearly with phosphors at neutral pH. blend[2]. |
| References |
[1]. Caspase activation by anticancer drugs: the caspase storm. Mol Pharm. 2007 Jul-Aug;4(4):583-95. [2]. A continuous fluorometric assay for gamma-glutamyltranspeptidase. Anal Biochem. 1996 Jan 1;233(1):71-5. |
| Additional Infomation | 7-amino-4-(trifluoromethyl)coumarin is a member of 7-aminocoumarins. It has a role as a fluorochrome. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~436.38 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (10.91 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.3638 mL | 21.8188 mL | 43.6376 mL | |
| 5 mM | 0.8728 mL | 4.3638 mL | 8.7275 mL | |
| 10 mM | 0.4364 mL | 2.1819 mL | 4.3638 mL |