ACTB-1003 is an orally available multi-kinase inhibitor with IC50 values of 6, 2 and 4 nM for FGFR1, VEGFR2 and Tie-2. It inhibits VEGFR2 and Tie-2, targets cancer mutations (FGFR inhibition), and induces apoptosis (by targeting RSK and p70S6K, which are downstream of PI3 kinase). Due to its multi-activity, ACTB-1003 has been shown to be effective in vivo against a range of histological cancers, including colorectal, lung, and breast cancers, showing dose-dependent inhibition of tumor growth. OPM2 human multiple myeloma and the murine leukemia Ba/F3-TEL-FGFR1 are two cell lines with FGFR genetic alterations in which ACTB-1003 is highly active with dose-dependent tumor growth inhibition. FGFR3 t(4:14) translocation, FGFR3 K650E mutation, and PTEN deletion are present in OPM2 cells, whereas FGFR1 over-expression drives Ba/F3-TEL-FGFR1 cells.
Physicochemical Properties
| Molecular Formula | C27H26F5N7O3 | |
| Molecular Weight | 591.53 | |
| Exact Mass | 591.202 | |
| Elemental Analysis | C, 54.82; H, 4.43; F, 16.06; N, 16.58; O, 8.11 | |
| CAS # | 939805-30-8 | |
| Related CAS # |
|
|
| PubChem CID | 23653175 | |
| Appearance | White to off-white solid powder | |
| Density | 1.51±0.1 g/cm3 (20 °C, 760 mmHg) | |
| LogP | 5.563 | |
| Hydrogen Bond Donor Count | 3 | |
| Hydrogen Bond Acceptor Count | 12 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 42 | |
| Complexity | 901 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O=C(NC1C(F)=CC=C(C(F)(F)F)C=1)NC1C(F)=CC(C2C(COC)=C(CN3CCOCC3)N3C=2C(N)=NC=N3)=CC=1 |
|
| InChi Key | GZPJCJKUZPUFAL-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C27H26F5N7O3/c1-41-13-17-22(12-38-6-8-42-9-7-38)39-24(25(33)34-14-35-39)23(17)15-2-5-20(19(29)10-15)36-26(40)37-21-11-16(27(30,31)32)3-4-18(21)28/h2-5,10-11,14H,6-9,12-13H2,1H3,(H2,33,34,35)(H2,36,37,40) | |
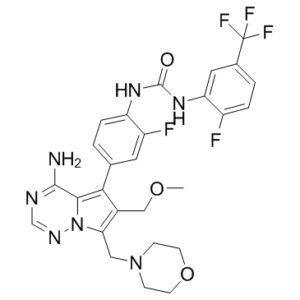
| Chemical Name | 1-[4-[4-amino-6-(methoxymethyl)-7-(morpholin-4-ylmethyl)pyrrolo[2,1-f][1,2,4]triazin-5-yl]-2-fluorophenyl]-3-[2-fluoro-5-(trifluoromethyl)phenyl]urea | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
FGFR1 (IC50 = 6 nM); VEGFR2 (IC50 = 2 nM); Tie-2 (IC50 = 4 nM)
ACTB-1003: Fibroblast growth factor receptor (FGFR, including mutant forms) (IC50=12 nM for FGFR1, IC50=18 nM for FGFR2, IC50=21 nM for FGFR3 G380R mutant) [1] ACTB-1003: Vascular endothelial growth factor receptor 2 (VEGFR2/KDR) (IC50=9 nM) [1] ACTB-1003: TEK (Tie2) (IC50=25 nM) [1] ACTB-1003: Ribosomal S6 kinase (RSK) (IC50=32 nM) [1] ACTB-1003: p70S6K (IC50=28 nM) [1] ACTB-1003 exhibited >50-fold selectivity for the above kinases over other tyrosine and serine/threonine kinases (e.g., EGFR, PDGFR, AKT) at concentrations up to 1 μM [1] |
| ln Vitro |
EOC317 (ACTB-1003) is an oral kinase inhibitor that functions in several ways. It targets cancer mutations by inhibiting FGFR1 (IC50=6 nM), angiogenesis by inhibiting VEGFR2 (2 nM), Tie-2 (4 nM), and likely apoptosis by inhibiting RSK (5 nM) and p70S6K (32\M). In cell lines with FGFR genetic alterations—OPM2 human multiple myeloma and the murine leukemia Ba/F3-TEL-FGFR1—EOC317 is highly active and dose-dependently inhibits tumor growth. Ba/F3-TEL-FGFR1 cells are driven by FGFR1 over-expression, whereas OPM2 cells have the FGFR3 t(4:14) translocation, FGFR3 K650E mutation, and PTEN deletion[1]. 1. ACTB-1003 potently inhibited the kinase activity of recombinant FGFR (1-3), VEGFR2, TEK, RSK, and p70S6K with IC50 values ranging from 9 to 32 nM, and showed minimal activity against off-target kinases (IC50 >1 μM for EGFR, PDGFRβ, c-Met) [1] 2. In human cancer cell lines harboring FGFR mutations (NCI-H1581, FGFR1 amplification; SNU-16, FGFR2 amplification; RT112, FGFR3 mutation), ACTB-1003 induced dose-dependent growth inhibition with EC50 values of 45 nM, 62 nM, and 58 nM, respectively [1] 3. In human umbilical vein endothelial cells (HUVECs), ACTB-1003 inhibited VEGF-induced proliferation and tube formation with IC50 values of 35 nM and 42 nM, confirming its anti-angiogenic activity via VEGFR2 inhibition [1] 4. ACTB-1003 (50 nM) induced apoptosis in FGFR-mutant cancer cells (NCI-H1581) by 40% after 72 hours of treatment, as measured by Annexin V/PI staining; this effect was associated with cleavage of caspase-3 and PARP (detected by western blot) [1] 5. Western blot analysis showed that ACTB-1003 (10-100 nM) dose-dependently reduced phosphorylation of FGFR downstream targets (ERK1/2, AKT) and RSK/p70S6K substrates (S6 ribosomal protein) in cancer cells, confirming blockade of the FGFR and mTOR/RSK signaling pathways [1] 6. ACTB-1003 suppressed clonogenic growth of FGFR-amplified cancer cells (NCI-H1581) with a 75% reduction in colony formation at 100 nM, compared to vehicle-treated controls [1] |
| ln Vivo |
EOC317 (ACTB-1003) reduces the growth of new blood vessels in tumor sections, as evidenced by the suppression of CD31 staining. Based on the HCT-116 colon tumor xenograft model, EOC317 can be combined with 5-FU or paclitaxel without reducing their efficacy or making them more toxic[1]. 1. In NCI-H1581 (FGFR1-amplified) lung cancer xenografts in nude mice, oral administration of ACTB-1003 (50, 100, 150 mg/kg/day) caused dose-dependent tumor growth inhibition, with a 68% reduction in tumor volume at 100 mg/kg/day after 21 days of treatment; the 150 mg/kg/day dose achieved complete tumor stasis [1] 2. In RT112 (FGFR3-mutant) bladder cancer xenografts, ACTB-1003 (100 mg/kg/day, oral) inhibited tumor growth by 62% and reduced microvessel density (CD31 staining) by 55% in tumor tissues, demonstrating anti-angiogenic effects in vivo [1] 3. Pharmacodynamic analysis of tumor tissues from treated mice showed that ACTB-1003 (100 mg/kg) reduced phospho-FGFR, phospho-VEGFR2, and phospho-S6 levels by 70-80% at 4 hours post-administration, confirming target engagement in vivo [1] 4. In a murine Matrigel plug angiogenesis model, ACTB-1003 (50 mg/kg/day, oral) reduced vascularization of Matrigel plugs by 60% compared to vehicle controls, with a significant decrease in hemoglobin content (a marker of blood vessel formation) [1] 5. Mice treated with ACTB-1003 (100 mg/kg/day for 28 days) showed no significant weight loss or signs of systemic toxicity, and histopathological examination of major organs (liver, kidney, heart) revealed no drug-related lesions [1] |
| Enzyme Assay |
1. Recombinant kinase activity assay for FGFR/VEGFR2/TEK [1] : Purified recombinant human FGFR1, FGFR2, FGFR3 (G380R mutant), VEGFR2, and TEK kinases were incubated with serial dilutions of ACTB-1003 (0.1 nM-1 μM) in kinase reaction buffer containing ATP (100 μM) and a fluorescent peptide substrate specific to each kinase. The reaction was incubated at 30°C for 1 hour, and kinase activity was measured by quantifying phosphorylated substrate fluorescence (excitation 485 nm, emission 535 nm). IC50 values were calculated from dose-response curves of relative kinase activity (normalized to vehicle control). 2. RSK/p70S6K kinase activity assay [1] : Recombinant human RSK1 and p70S6K were incubated with ACTB-1003 (0.1 nM-1 μM) in reaction buffer with [γ-³²P]ATP and a synthetic peptide substrate (S6 peptide for p70S6K, CREB peptide for RSK). After 30 minutes at 37°C, the reaction was spotted onto phosphocellulose paper, unbound radioactivity was washed away, and incorporated ³²P was quantified by scintillation counting. IC50 values were determined from the inhibition of radioactive phosphate incorporation. 3. Kinase selectivity panel assay [1] : ACTB-1003 (1 μM) was tested against a panel of 80 human kinases (tyrosine and serine/threonine kinases) using the same kinase activity assay conditions as above. The percentage of kinase inhibition was calculated for each target, and selectivity was defined as >50-fold higher IC50 for off-target kinases compared to the primary targets (FGFR, VEGFR2, TEK, RSK, p70S6K). |
| Cell Assay |
1. Cancer cell proliferation assay [1] : Human cancer cell lines (NCI-H1581, SNU-16, RT112, A549) were seeded in 96-well plates at a density of 2×10³ cells/well and treated with serial dilutions of ACTB-1003 (1 nM-10 μM) for 72 hours. Cell viability was measured using a colorimetric MTT assay, and EC50 values for growth inhibition were calculated from dose-response curves. Non-FGFR-mutant cell lines (A549) were used as negative controls to confirm target specificity. 2. HUVEC proliferation and tube formation assay [1] : HUVECs were seeded in 96-well plates (5×10³ cells/well) and treated with ACTB-1003 (1 nM-1 μM) in the presence of VEGF (20 ng/mL) for 48 hours; cell viability was assessed by MTT assay to determine the IC50 for anti-proliferative activity. For tube formation, HUVECs were seeded on Matrigel-coated 24-well plates and treated with ACTB-1003 (10 nM-1 μM) plus VEGF (20 ng/mL); tube formation was photographed after 6 hours, and the number of capillary-like structures was quantified using image analysis software. 3. Apoptosis and western blot analysis [1] : NCI-H1581 cells were treated with ACTB-1003 (10, 50, 100 nM) for 48 and 72 hours, then stained with Annexin V-FITC and propidium iodide (PI) for flow cytometric analysis of apoptosis. For western blotting, cell lysates were prepared, and equal amounts of protein were separated by SDS-PAGE, transferred to membranes, and probed with antibodies against phospho-FGFR, total FGFR, phospho-ERK1/2, phospho-AKT, cleaved caspase-3, PARP, and GAPDH (loading control). Band intensities were quantified using imaging software to measure pathway inhibition and apoptotic marker expression. 4. Colony-forming assay [1] : NCI-H1581 cells were seeded in 6-well plates at a density of 500 cells/well in soft agar medium containing ACTB-1003 (10, 50, 100 nM) or vehicle. Colonies were allowed to form for 14 days at 37°C with 5% CO₂, then stained with crystal violet and counted under a microscope. The percentage of colony formation was calculated relative to the vehicle control group. |
| Animal Protocol |
HCT-116 colon tumor xenograft model. 1. Xenograft tumor model (NCI-H1581 and RT112) [1] : Female nude mice (6-8 weeks old) were injected subcutaneously with NCI-H1581 (5×10⁶ cells/mouse) or RT112 (1×10⁷ cells/mouse) cancer cells into the right flank. When tumors reached a volume of 100-150 mm³, mice were randomized into treatment groups (vehicle, 50, 100, 150 mg/kg ACTB-1003) and dosed orally once daily for 21 days. ACTB-1003 was formulated as a suspension in 0.5% methylcellulose/0.1% Tween 80. Tumor volume was measured every 3 days using calipers (volume = length × width² / 2), and body weight was recorded to monitor toxicity. 2. Matrigel plug angiogenesis model [1] : C57BL/6 mice were injected subcutaneously with Matrigel (0.5 mL/mouse) containing VEGF (50 ng/mL) and heparin (10 U/mL) into the ventral region. ACTB-1003 (50 mg/kg/day) or vehicle was administered orally daily for 7 days. Matrigel plugs were excised, weighed, and homogenized to measure hemoglobin content using a colorimetric assay (Drabkin’s reagent) as an indicator of vascularization. 3. Pharmacodynamic analysis in xenografts [1] : Mice bearing NCI-H1581 xenografts were dosed orally with ACTB-1003 (100 mg/kg), and tumor tissues were collected at 2, 4, 8, and 24 hours post-administration. Tumor lysates were prepared, and western blot analysis was performed to measure phospho-FGFR, phospho-VEGFR2, and phospho-S6 levels. Plasma samples were also collected to determine ACTB-1003 concentrations by LC-MS/MS. |
| ADME/Pharmacokinetics |
1. ACTB-1003 had an oral bioavailability of 58% in mice, with a peak plasma concentration (Cmax) of 1.2 μM and an area under the curve (AUC₀-24h) of 9.8 μM·h following a single oral dose of 100 mg/kg [1] 2. The elimination half-life (t₁/₂) of ACTB-1003 in mice was 6.2 hours, and the drug showed good tissue distribution, with a tumor/plasma concentration ratio of 1.8 at 4 hours post-administration [1] 3. ACTB-1003 was metabolized primarily by hepatic CYP3A4 in human liver microsomes, with an intrinsic clearance of 15 μL/min/mg protein; it was not a substrate for P-glycoprotein (P-gp) [1] 4. The plasma protein binding of ACTB-1003 was 91% in mouse plasma and 94% in human plasma, with no concentration-dependent binding over the range of 0.1-10 μM [1] |
| Toxicity/Toxicokinetics |
1. In acute toxicity studies in mice, the oral LD50 of ACTB-1003 was >500 mg/kg, and the intraperitoneal LD50 was >200 mg/kg, indicating low acute toxicity [1] 2. Repeated oral administration of ACTB-1003 (100 mg/kg/day for 28 days) in rats did not cause significant changes in body weight, food intake, or clinical chemistry parameters (ALT, AST, creatinine, BUN); no histopathological abnormalities were observed in the liver, kidney, heart, or bone marrow [1] 3. ACTB-1003 did not inhibit major CYP450 enzymes (CYP3A4, CYP2D6, CYP2C9) at concentrations up to 10 μM, suggesting a low risk of drug-drug interactions [1] 4. In hematological toxicity assessments, ACTB-1003 (100 mg/kg/day for 21 days) did not reduce peripheral blood leukocyte, red blood cell, or platelet counts in mice [1] 5. The most common dose-limiting toxicity in preclinical studies was mild diarrhea at doses >150 mg/kg/day in mice, which was reversible upon dose reduction [1] |
| References |
[1]. ACTB-1003: An oral kinase inhibitor targeting cancer mutations (FGFR), angiogenesis (VEGFR2, TEK), and induction of apoptosis (RSK and p70S6K). Journal of Clinical Oncology 28, no. 15 DOI: 10.1200/jco.2010.28.15_suppl.e13665. |
| Additional Infomation |
Multi-mode Kinase Inhibitor EOC317 is an orally available, small molecule, multi-mode kinase inhibitor (MMKI), with potential antineoplastic activity. Upon oral administration, MMKI EOC317 targets, binds to and inhibits the activity of a variety of kinases, such as phosphatidylinositol 3 kinase (PI3K), and the receptor tyrosine kinases, fibroblast growth factor receptor (FGFR), angiopoietin-1 receptor (TIE 2), and vascular endothelial growth factor receptor-2 (VEGFR-2). This inhibition may result in an induction of apoptosis of susceptible tumors cells in which these kinases are overexpressed. 1. ACTB-1003 is an oral, multi-targeted small-molecule kinase inhibitor designed to simultaneously target cancer-driving FGFR mutations, tumor angiogenesis (VEGFR2/TEK), and pro-survival signaling pathways (RSK/p70S6K) [1] 2. The dual mechanism of ACTB-1003 (direct anti-tumor activity via FGFR inhibition and anti-angiogenic activity via VEGFR2/TEK inhibition) provides a synergistic approach to cancer treatment, particularly for FGFR-amplified or mutant solid tumors [1] 3. At the time of publication, ACTB-1003 was being evaluated in a Phase I clinical trial (NCT01226146) for patients with advanced solid tumors harboring FGFR alterations (amplifications, mutations, or fusions) [1] 4. The Phase I trial of ACTB-1003 aimed to determine the maximum tolerated dose (MTD), safety profile, pharmacokinetics, and preliminary anti-tumor activity in patients with refractory solid cancers (lung, bladder, gastric, breast cancer) [1] 5. ACTB-1003 is distinguished from other FGFR inhibitors by its additional activity against RSK and p70S6K, which are key mediators of cancer cell survival and resistance to targeted therapy [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6905 mL | 8.4527 mL | 16.9053 mL | |
| 5 mM | 0.3381 mL | 1.6905 mL | 3.3811 mL | |
| 10 mM | 0.1691 mL | 0.8453 mL | 1.6905 mL |