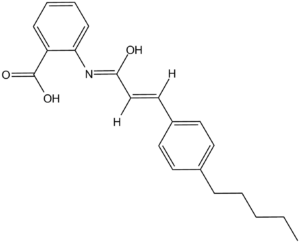
ACA [also known as N-(p-amylcinnamoyl) Anthranilic Acid] is a broad-spectrum inhibitor of phospholipase A2 (PLA2) and a TRP channel blocker. It is structurally-related to non-steroidal anti-inflammatory drugs (NSAIDs) of the fenamate group and may also modulate various ion channels. In HEK293 cells transfected with human TRPM2, ACA (20 µM) completely blocked H2O2-induced Ca2+ signals and ADPR-induced whole-cell currents with IC50 value of 1.7 µM. The inhibitory effects of ACA on I(Cl(Ca)) are PLA(2)-independent and that the drug may serve as a useful tool in understanding the nature and function of cardiac anion channels.
Physicochemical Properties
| Molecular Formula | C21H23NO3 | |
| Molecular Weight | 337.41 | |
| Exact Mass | 337.167 | |
| CAS # | 110683-10-8 | |
| Related CAS # |
|
|
| PubChem CID | 5353376 | |
| Appearance | White to off-white solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 563.1±50.0 °C at 760 mmHg | |
| Flash Point | 294.4±30.1 °C | |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C | |
| Index of Refraction | 1.629 | |
| LogP | 7.11 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 8 | |
| Heavy Atom Count | 25 | |
| Complexity | 452 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | CCCCCC1=CC=C(C=C1)/C=C/C(=O)NC2=CC=CC=C2C(=O)O |
|
| InChi Key | GAMRBCZMOOMBSQ-CCEZHUSRSA-N | |
| InChi Code | InChI=1S/C21H23NO3/c1-2-3-4-7-16-10-12-17(13-11-16)14-15-20(23)22-19-9-6-5-8-18(19)21(24)25/h5-6,8-15H,2-4,7H2,1H3,(H,22,23)(H,24,25)/b15-14+ | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | N-(p-amylcinnamoyl) Anthranlilic Acid (ACA; 20 μM) totally inhibits H2O2-induced Ca2+ signals and ADPR-induced whole-cell currents in HEK293 cells transfected with human TRPM2 (IC50=1.7 μM) [1]. Additionally, N-(p-amylcinnamoyl) Anhranilic Acid (ACA; 20 μM) burns current through human TRPM8 and TRPC6[1], which are expressed in HEK293 cells. Several TRP channels are regulated by N-(p-amylcinnamoyl) Anthranlilic Acid (ACA) separately from PLA22 inhibition [1]. |
| ln Vitro | N-(p-amylcinnamoyl) Anthranlilic Acid (ACA; 20 μM) totally inhibits H2O2-induced Ca2+ signals and ADPR-induced whole-cell currents in HEK293 cells transfected with human TRPM2 (IC50=1.7 μM) [1]. Additionally, N-(p-amylcinnamoyl) Anhranilic Acid (ACA; 20 μM) burns current through human TRPM8 and TRPC6[1], which are expressed in HEK293 cells. Several TRP channels are regulated by N-(p-amylcinnamoyl) Anthranlilic Acid (ACA) separately from PLA22 inhibition [1]. |
| Enzyme Assay |
The provided text does not describe specific enzyme activity assays for ACA. The study focuses on its ion channel blocking activity. [1] |
| Cell Assay |
Whole-cell Patch-Clamp Recording: Membrane currents were measured in the whole-cell configuration of the patch-clamp technique. Cells were placed in a recording chamber perfused with bath solution. Pipettes were filled with an intracellular solution. For TRPM2 activation, the pipette solution contained 1 mM ADPR. For TRPC6 activation, the pipette solution contained 30 µM AlF4-. Currents were elicited by voltage ramps from -100 to +100 mV applied every 2 seconds. To test drug effects, ACA or other compounds were applied extracellularly via the bath perfusion. Currents were recorded, filtered, digitized, and analyzed. [1] Calcium Imaging (Single Cells): Intracellular Ca2+ concentration ([Ca2+]i) in single TRPM2-transfected HEK293 cells was measured using the fluorescent indicator fura-2. Cells were loaded with fura-2-AM. Fluorescence was excited at 340 nm and 380 nm. The fluorescence ratio F340/F380 was calculated after background subtraction to monitor changes in [Ca2+]i. H2O2 was used as an extracellular activator of TRPM2, and the effect of pre-application of ACA was tested. [1] Calcium Imaging (Plate Reader): [Ca2+]i measurements in TRPM8-transfected HEK293 cells grown in 96-well plates were performed using the fluorescent indicator fluo-4. Cells were loaded with fluo-4. After loading, the solution was exchanged for a Ca2+-free solution containing EGTA. The plate was inserted into a fluorometric imaging plate reader. Fluorescence was excited at 485 nm and emission measured at 535 nm. Menthol, ACA, and CaCl2 were added consecutively to the wells, and fluorescence changes were monitored. [1] Western Blot: Membrane fractions from various cell lines were separated by SDS-PAGE on a 6% polyacrylamide gel and transferred to nitrocellulose membranes. Membranes were incubated with a primary antibody against TRPM2 overnight at 4°C. After washing, bound antibody was detected using a chemiluminescence detection kit to confirm TRPM2 protein expression. [1] |
| References |
[1]. Inhibition of TRPM2 cation channels by N-(p-amylcinnamoyl)anthranilic acid. Br J Pharmacol. 2006 Jun;148(3):264-73. [2]. N-(p-amylcinnamoyl)anthranilic acid (ACA): a phospholipase A(2) inhibitor and TRP channel blocker. Cardiovasc Drug Rev. 2007 Spring;25(1):61-75. [3]. Inhibition of the calcium-activated chloride current in cardiac ventricular myocytes by N-(p-amylcinnamoyl)anthranilic acid (ACA). Biochem Biophys Res Commun. 2010 Nov 19;402(3):531-6. |
| Additional Infomation |
N-(p-amylcinnamoyl)anthranilic acid is an amidobenzoic acid that is anthranilic acid in which one of the anilino hydrogens is replaced by a 4-pentylcinnamoyl group. It is a transient receptor potential (TRP) channel blocker and phospholipase A2 (PLA2) inhibitor. It has a role as an EC 3.1.1.4 (phospholipase A2) inhibitor and a TRP channel blocker. It is an amidobenzoic acid, a member of cinnamamides and a secondary carboxamide. ACA (N-(p-amylcinnamoyl)anthranilic acid) is structurally an N-cinnamoylanthranilic acid derivative. It was previously known as a phospholipase A2 inhibitor. This study identifies it as a direct modulator of several TRP channels (TRPM2, TRPM8, TRPC6). The inhibition of TRPM2 by ACA is proposed to occur via an extracellular site, modulating channel gating. The block is pH-dependent, being faster at lower pH, which is consistent with ACA being a weak acid. Due to its potency, ACA can serve as a useful pharmacological tool, in combination with other blockers, to study the function of TRPM2 and related channels in native cells. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.16 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.16 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9638 mL | 14.8188 mL | 29.6375 mL | |
| 5 mM | 0.5928 mL | 2.9638 mL | 5.9275 mL | |
| 10 mM | 0.2964 mL | 1.4819 mL | 2.9638 mL |