Physicochemical Properties
| Molecular Formula | C18H31NAO3 |
| Molecular Weight | 318.4328 |
| Exact Mass | 296.234 |
| CAS # | 57818-44-7 |
| PubChem CID | 21158511 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.0±0.0 g/cm3 |
| Boiling Point | 409.6±0.0 °C at 760 mmHg |
| Flash Point | 215.7±0.0 °C |
| Vapour Pressure | 0.0±0.0 mmHg at 25°C |
| Index of Refraction | 1.492 |
| LogP | 6.24 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 21 |
| Complexity | 295 |
| Defined Atom Stereocenter Count | 0 |
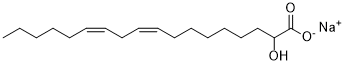
| SMILES | CCCCC/C=C\C/C=C\CCCCCCC(O)C(O)=O |
| InChi Key | VFXKYDDSDQXKLC-NBTZWHCOSA-M |
| InChi Code | InChI=1S/C18H32O3.Na/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17(19)18(20)21/h6-7,9-10,17,19H,2-5,8,11-16H2,1H3,(H,20,21)/q+1/p-1/b7-6-,10-9- |
| Chemical Name | sodium (9Z,12Z)-2-hydroxyoctadeca-9,12-dienoate |
| Synonyms | ABTL0812 ABTL 0812ABTL-0812 sodium 2-hydroxylinoleate. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Squamous NSCLC H157 cells' capacity to proliferate is inhibited by ABTL-0812 (ABTL0812; 10-100 μM; 48 hours) [1]. Treatment with ABTL0812 does not affect the human lung fibroblast cell line MRC-5 as it does not affect squamous non-small cell lung carcinoma H157 cells [1]. |
| ln Vivo | In human lung and pancreas xenografts, ABTL-0812 (ABTL0812; 120 mg/kg; oral gavage; 5 times per week; for 33 days) causes ER stress [1]. In vivo ER stress signals are induced by ABTL-0812. ATF4 and HSPA5 expression is upregulated in mice bearing A549 and MiaPaca2 xenografts, respectively, by ABTL-0812 [1]. |
| Cell Assay |
Cell Viability Assay [1] Cell Types: Human lung fibroblasts MRC5 and squamous non-small cell lung (NSCLC) H157 cells Tested Concentrations: 10, 30, 100 μM Incubation Duration: 48 hrs (hours) Experimental Results: Inhibition of squamous NSCLC H157 cells viability, but not the lung fibroblast cell line MRC-5. |
| Animal Protocol |
Animal/Disease Models: MiaPaca2 and A549 xenograft models in athymic female nude mice [1] Doses: 120 mg/kg Route of Administration: po (oral gavage); 5 times per week for 33 days Experimental Results: In human lung and pancreas xenografts Inducing ER stress. |
| References |
[1]. The anti-cancer drug ABTL0812 induces ER stress-mediated cytotoxic autophagy by increasing dihydroceramide levels in cancer cells. Autophagy. 2020 May 13. |
| Additional Infomation |
2-hydroxylinoleic acid is a 2-hydroxy fatty acid derived from linoleic acid. It has a role as an Arabidopsis thaliana metabolite, an antineoplastic agent, a PPARalpha agonist and a PPARgamma agonist. It is a 2-hydroxy fatty acid, a long-chain fatty acid and a HODE. It is functionally related to a linoleic acid. alpha-Hydroxylinoleic acid is under investigation in clinical trial NCT04431258 (ABTL0812 in Combination With FOLFIRINOX for First-line Treatment of Metastatic Pancreatic Study). Ibrilatazar is an orally bioavailable, lipid analogue and inhibitor of raptor-mammalian target of rapamycin (mTOR) (mTOR complex 1; mTORC1), rictor-mTOR (mTOR complex 2; mTORC2) and dihydrofolate reductase (DHFR) with potential antineoplastic activity. Upon oral administration, ibrilatazar binds to and inhibits both mTORC1 and mTORC2, which may result in apoptosis and a decrease in proliferation in mTORC1/2-expressing tumor cells. mTOR is a serine/threonine kinase that is upregulated in some tumors; it plays an important role in the PI3K/Akt/mTOR signaling pathway which is often deregulated in cancer cells. In addition, ibrilatazar inhibits DHFR, an enzyme that reduces dihydrofolic acid to tetrahydrofolic acid, thereby blocking tetrahydrofolate synthesis, and resulting in both the depletion of nucleotide precursors and the inhibition of DNA, RNA and protein synthesis. This induces autophagy-induced cell death and further inhibition of cell proliferation. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~843.34 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (7.02 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (7.02 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (7.02 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.1404 mL | 15.7020 mL | 31.4041 mL | |
| 5 mM | 0.6281 mL | 3.1404 mL | 6.2808 mL | |
| 10 mM | 0.3140 mL | 1.5702 mL | 3.1404 mL |