|
ABT-639 hydrochloride (HCl) is a new potent, peripherally acting, selective T-type Ca2+ channel blocker that blocks recombinant human T-type (Cav3.2) Ca2+ channels in a voltage-dependent fashion (IC50=2 μM) and attenuates low voltage-activated (LVA) currents in rat DRG neurons (IC50=8 μM). ABT-639 is significantly less active at other Ca2+ channels (e.g. Cav1.2 and Cav2.2) (IC50>30 mM). Following oral administration ABT-639 produced dose-dependent antinociception in a rat model of knee joint pain (ED₅₀ = 2 mg/kg, p.o.). ABT-639 (10-100 mg/kg, p.o.) also increased tactile allodynia thresholds in multiple models of neuropathic pain (e.g. spinal nerve ligation, CCI, and vincristine-induced). [corrected]. ABT-639 did not attenuate hyperalgesia in inflammatory pain models induced by complete Freund's adjuvant or carrageenan. At higher doses (e.g. 100-300 mg/kg) ABT-639 did not significantly alter hemodynamic or psychomotor function. The antinociceptive profile of ABT-639 provides novel insights into the role of peripheral T-type (Ca(v)3.2) channels in chronic pain states.
|
Physicochemical Properties
| Molecular Formula |
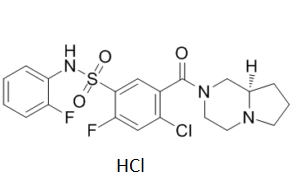
C₂₀H₂₁CL₂F₂N₃O₃S
|
| Molecular Weight |
492.37
|
| Exact Mass |
491.064
|
| CAS # |
1235560-31-2
|
| Related CAS # |
ABT-639;1235560-28-7
|
| PubChem CID |
122180940
|
| Appearance |
Typically exists as solid at room temperature
|
| Hydrogen Bond Donor Count |
2
|
| Hydrogen Bond Acceptor Count |
7
|
| Rotatable Bond Count |
4
|
| Heavy Atom Count |
31
|
| Complexity |
737
|
| Defined Atom Stereocenter Count |
1
|
| SMILES |
O=S(C1=CC(C(N2C[C@](CCC3)([H])N3CC2)=O)=C(Cl)C=C1F)(NC4=CC=CC=C4F)=O.Cl
|
| InChi Key |
KZRCYRFDOFTBHY-BTQNPOSSSA-N
|
| InChi Code |
InChI=1S/C20H20ClF2N3O3S.ClH/c21-15-11-17(23)19(30(28,29)24-18-6-2-1-5-16(18)22)10-14(15)20(27)26-9-8-25-7-3-4-13(25)12-26;/h1-2,5-6,10-11,13,24H,3-4,7-9,12H2;1H/t13-;/m1./s1
|
| Chemical Name |
5-[(8aR)-3,4,6,7,8,8a-hexahydro-1H-pyrrolo[1,2-a]pyrazine-2-carbonyl]-4-chloro-2-fluoro-N-(2-fluorophenyl)benzenesulfonamide;hydrochloride
|
| Synonyms |
| ABT-639 HCl; ABT 639; ABT639 |
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder-20°C 3 years
4°C 2 years
In solvent -80°C 6 months
-20°C 1 month
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
|
Biological Activity
| ln Vitro |
In vitro activity: ABT-639 is a new potent, peripherally acting, selective T-type Ca2+ channel blocker that blocks recombinant human T-type (Cav3.2) Ca2+ channels in a voltage-dependent fashion (IC50=2 μM) and attenuates low voltage-activated (LVA) currents in rat DRG neurons (IC50=8 μM). ABT-639 is significantly less active at other Ca2+ channels (e.g. Cav1.2 and Cav2.2) (IC50>30 mM). Following oral administration ABT-639 produced dose-dependent antinociception in a rat model of knee joint pain (ED₅₀ = 2 mg/kg, p.o.). ABT-639 (10-100 mg/kg, p.o.) also increased tactile allodynia thresholds in multiple models of neuropathic pain (e.g. spinal nerve ligation, CCI, and vincristine-induced). [corrected]. ABT-639 did not attenuate hyperalgesia in inflammatory pain models induced by complete Freunds adjuvant or carrageenan. At higher doses (e.g. 100-300 mg/kg) ABT-639 did not significantly alter hemodynamic or psychomotor function. The antinociceptive profile of ABT-639 provides novel insights into the role of peripheral T-type (Ca(v)3.2) channels in chronic pain states.
Kinase Assay:
Cell Assay: ABT-639 blocks recombinant human T-type (Cav3.2) Ca2+ channels in a voltage-dependent fashion (IC50=2 μM) and attenuates low voltage-activated (LVA) currents in rat DRG neurons (IC50=8 μM). ABT-639 is significantly less active at other Ca2+ channels (e.g. Cav1.2 and Cav2.2) (IC50>30 mM). ABT-639 has high oral bioavailability (%F=73), low protein binding (88.9%) and a low brain:plasma ratio (0.05:1) in rodents. Following oral administration |
|
| ln Vivo |
ABT-639 attenuates low voltage-activated (LVA) currents in rat DRG neurons (IC50=8 μM) and inhibits recombinant human T-type (Cav3.2) Ca2+ channels in a voltage-dependent manner (IC50=2 μM). ABT -639 exhibits much lower activity in other Ca2+ channels (IC50>30 mM), such as Cav1.2 and Cav2.2. In animals, ABT-639 exhibits low protein binding (88.9%), a low brain:plasma ratio (0.05:1), and good oral bioavailability (%F=73). In a rat model of knee joint discomfort, oral treatment of ABT-639 results in dose-dependent antinociception (ED50=2 mg/kg, po). In several models of neuropathic pain, such as spinal nerve ligation, CCI, vincristine-induced, and capsaicin secondary hypersensitivity, ABT-639 (10-100 mg/kg, po) also raises tactile allodynia thresholds. In inflammatory pain models generated by carrageenan or full Freund's adjuvant, ABT-639 did not reduce hyperalgesia. Higher doses of ABT-639 (such as 100–300 mg/kg) have no discernible effects on hemodynamic or psychomotor performance. Novel insights into the function of peripheral T-type (Cav3.2) channels in chronic pain conditions are offered by the antinociceptive characteristics of ABT-639[1].
|
| Animal Protocol |
| 10-100 mg/kg, p.o. | | Rat model of knee joint pain | |
|
| References |
[1]. Jarvis MF, et al. A peripherally acting, selective T-type calcium channel blocker, ABT-639, effectively reduces nociceptive and neuropathic pain in rats. Biochem Pharmacol. 2014 Jun 15;89(4):536-44
|
|
Solubility Data
| Solubility (In Vitro) |
| DMSO:10 mg/mL | | Water:<1 mg/mL | | Ethanol:<1 mg/mL |
|
| Solubility (In Vivo) |
| O=S(C1=CC(C(N2C[C@](CCC3)([H])N3CC2)=O)=C(Cl)C=C1F)(NC4=CC=CC=C4F)=O.Cl |
(Please use freshly prepared in vivo formulations for optimal results.)
|
| Preparing Stock Solutions |
|
1 mg |
5 mg |
10 mg |
| 1 mM |
2.0310 mL |
10.1550 mL |
20.3099 mL |
| 5 mM |
0.4062 mL |
2.0310 mL |
4.0620 mL |
| 10 mM |
0.2031 mL |
1.0155 mL |
2.0310 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles. |