|
A32390A, an isonitrile-containing analog of mannitol, is a new class of antifungal antibiotics. In vitro antifungal activity of A32390A was found against Candida albicans, Cryptococcus neoformans and Histoplasma capsulatum. In vivo antifungal activity of A32390A was observed in mice infected with C. albicans. Accumulative doses of 37.5 approximately 600 mg/kg, administered subcutaneously over a 24-hour period, showed significant activity without demonstrating toxicity. A32390A was effective, but not as effective as amphotericin B, in reducing the number of Candida cells isolated from the kidney of infected mice.
|
Physicochemical Properties
| Molecular Formula |
C18H24N2O8
|
| Molecular Weight |
396.39176
|
| Exact Mass |
396.153
|
| CAS # |
61241-59-6
|
| Related CAS # |
|
| PubChem CID |
43589
|
| Appearance |
Typically exists as solid at room temperature
|
| LogP |
0.3
|
| Hydrogen Bond Donor Count |
4
|
| Hydrogen Bond Acceptor Count |
10
|
| Rotatable Bond Count |
11
|
| Heavy Atom Count |
28
|
| Complexity |
680
|
| Defined Atom Stereocenter Count |
0
|
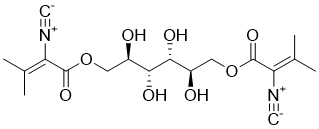
| SMILES |
[C-]#[N+]/C(/C(OCC(C(C(C(COC(/C(=C(/C)\C)/[N+]#[C-])=O)O)O)O)O)=O)=C(/C)\C
|
| InChi Key |
YEMJVHOPMVQUGS-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C18H24N2O8/c1-9(2)13(19-5)17(25)27-7-11(21)15(23)16(24)12(22)8-28-18(26)14(20-6)10(3)4/h11-12,15-16,21-24H,7-8H2,1-4H3
|
| Chemical Name |
[2,3,4,5-tetrahydroxy-6-(2-isocyano-3-methylbut-2-enoyl)oxyhexyl] 2-isocyano-3-methylbut-2-enoate
|
| Synonyms |
| A32390A; A-32390A; A 32390A |
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder-20°C 3 years
4°C 2 years
In solvent -80°C 6 months
-20°C 1 month
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
|
Biological Activity
| ln Vitro |
A32390A, an isonitrile-containing derivative of mannitol, represents a new class of antifungal antibiotics. In vitro antifungal activity of A32390A was found against Candida albicans, Cryptococcus neoformans and Histoplasma capsulatum. In vivo antifungal activity of A32390A was demonstrated in mice infected with C. albicans. Accumulative doses of 37.5 approximately 600 mg/kg, administered subcutaneously over a 24-hour period, showed significant activity without demonstrating toxicity. A32390A was effective, but not as effective as amphotericin B, in reducing the number of Candida cells isolated from the kidney of infected mice. Urinary excretion of A32390A accounted for only 10% of the administered dose. Improved bioavailability of A32390A was accomplished when the antibiotic was combined with polyvinyl pyrrolidone (PVP) in a solid dispersion. Administration of A32390A as a 10% dispersion in PVP resulted in increased urinary excretion of the drug and reduced the amount of drug required for in vivo activity.[1]
|
| References |
J Antibiot (Tokyo).1978 Jan;31(1):33-7.
|
| Additional Infomation |
Antibiotic A 32390A has been reported in Pyrenochaeta with data available.
|
|
Solubility Data
| Solubility (In Vitro) |
| DMSO:10 mM | | Water:N/A | | Ethanol:N/A |
|
| Solubility (In Vivo) |
| O[C@@H]([C@H]([C@@H]([C@@H](COC(/C([N+]#[C-])=C(C)\C)=O)O)O)O)COC(/C([N+]#[C-])=C(C)\C)=O |
(Please use freshly prepared in vivo formulations for optimal results.)
|
| Preparing Stock Solutions |
|
1 mg |
5 mg |
10 mg |
| 1 mM |
2.5228 mL |
12.6138 mL |
25.2277 mL |
| 5 mM |
0.5046 mL |
2.5228 mL |
5.0455 mL |
| 10 mM |
0.2523 mL |
1.2614 mL |
2.5228 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles. |