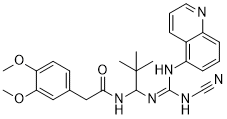
A-740003 is a potent and selective P2X7 receptor antagonist. A-740003 has (IC(50) values = 40 nM for human and 18 nM for rat) as measured by agonist-stimulated changes in intracellular calcium concentrations. A-740003 showed weak or no activity (IC(50) > 10 muM) at other P2 receptors and an array of other neurotransmitter and peptide receptors, ion channels, reuptake sites, and enzymes. A-740003 potently blocked agonist-evoked IL-1beta release (IC(50) = 156 nM) and pore formation (IC(50) = 92 nM) in differentiated human THP-1 cells. A-740003 in vivo produces significant antinociception in animal models of neuropathic and inflammatory pain.
Physicochemical Properties
| Molecular Formula | C26H30N6O3 |
| Molecular Weight | 474.565 |
| Exact Mass | 474.237 |
| CAS # | 861393-28-4 |
| Related CAS # | 861393-28-4; |
| PubChem CID | 23232014 |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Index of Refraction | 1.597 |
| LogP | 2.77 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 35 |
| Complexity | 773 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC(C)(C)C(NC(=O)CC1=CC(=C(C=C1)OC)OC)/N=C(/NC#N)\NC2=CC=CC3=C2C=CC=N3 |
| InChi Key | PUHSRMSFDASMAE-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C26H30N6O3/c1-26(2,3)24(31-23(33)15-17-11-12-21(34-4)22(14-17)35-5)32-25(29-16-27)30-20-10-6-9-19-18(20)8-7-13-28-19/h6-14,24H,15H2,1-5H3,(H,31,33)(H2,29,30,32) |
| Chemical Name | N-[1-[[(Cyanoamino)(5-quinolinylamino)methylene]amino]-2,2-dimethylpropyl]-3,4-dimethoxybenzeneacetamide |
| Synonyms | A-740003 A 740003 A740003. |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Significant boiling occurs in the sustained phase of the BzATP-sensing reaction when A-438079 or A-740003 (10 μM) is added [1]. Dentate granule cells with SE-conducted TNF-α expression are lessened upon infusion of A-740003. Neuronal death in SE sensors is increased by A-740003 infusion [2]. A-740003 and A-438079 showed noticeably higher potency in P2X7 receptor activation in all species when compared to other clamp antagonists. A-740003 and A-438079 show increased activity in storage and in people compared to the Muse P2X7 receptor [3]. In nursing human THP-1 cells, A-740003 potently inhibits agonist-induced IL-1β release (IC50=156 nM) and pore formation (IC50=92 nM) [4]. |
| ln Vivo | Systemic dosages of A-740003 demonstrated dose-dependent anti-injury effects in a tandem junctional nerve ligation paradigm (ED50=19 mg/kg ip). Additionally, sciatic nerve chronic ischemia and two other forms of neuropathic pain were lessened by A-740003. Furthermore, after carrageenan or Freund's adjuvant plantar model, A-740003 successfully and totally eliminated the heat hyperalgesia (ED50=38 - 54 mg/kg ip). A-740003 does not affect exercise performance at analgesic levels and is ineffective in decreasing normal state acute thermal nociceptors [4]. |
| References |
[1]. P2X?-mediated calcium influx triggers a sustained, PI3K-dependent increase in metabolic acid production by osteoblast-like cells. Am J Physiol Endocrinol Metab. 2012 Mar 1;302(5):E561-75. [2]. P2X7 receptor activation ameliorates CA3 neuronal damage via a tumor necrosis factor-α-mediated pathway in the rat hippocampus following status epilepticus. J Neuroinflammation. 2011 Jun 2;8:62. [3]. Mammalian P2X7 receptor pharmacology: comparison of recombinant mouse, rat and human P2X7 receptors.Br J Pharmacol. 2009 Aug;157(7):1203-14. Epub 2009 Jun 22. [4]. A-740003 [N-(1-{[(cyanoimino)(5-quinolinylamino) methyl]amino}-2,2-dimethylpropyl)-2-(3,4-dimethoxyphenyl)acetamide] a novel and selective P2X7 receptor antagonist, dose-dependently reduces neuropathic pain in the rat. J Pharmacol Exp Ther. 2006 Dec;319(3):1376-85. Epub 2006 Sep 18. [5]. Effect of electroacupuncture on the cervicospinal P2X7 receptor/fractalkine/CX3CR1 signaling pathway in a rat neck-incision pain model. Purinergic Signal. 2017 Jun;13(2):215-225. |
| Additional Infomation | See also: A-740003 (preferred). |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~105.36 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.27 mM) (saturation unknown) in 10% DMSO + 40% PEG300 +5% Tween-80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 + to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1072 mL | 10.5359 mL | 21.0717 mL | |
| 5 mM | 0.4214 mL | 2.1072 mL | 4.2143 mL | |
| 10 mM | 0.2107 mL | 1.0536 mL | 2.1072 mL |