A-484954 (A484954; A 484954) is a highly selective, cell-permeable and ATP-competitive eukaryotic elongation factor-2 (eEF2) inhibitor with potential antitumor activity. It inhibits eEF2 with an IC50 of 280 nM. eEF2 is a member of the GTP-binding translation elongation factor family, and an essential factor for protein synthesis and cell survival.
Physicochemical Properties
| Molecular Formula | C13H15N5O3 |
| Molecular Weight | 289.2899 |
| Exact Mass | 289.117 |
| CAS # | 142557-61-7 |
| PubChem CID | 14998470 |
| Appearance | White to off-white solid powder |
| Density | 1.522±0.06 g/cm3 (20 ºC 760 Torr) |
| LogP | 0.408 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 21 |
| Complexity | 492 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | HWODCHXORCTEGU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C13H15N5O3/c1-2-17-12(20)8-5-7(10(15)19)9(14)16-11(8)18(13(17)21)6-3-4-6/h5-6H,2-4H2,1H3,(H2,14,16)(H2,15,19) |
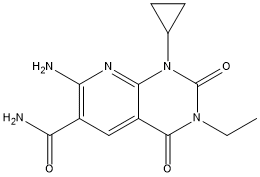
| Chemical Name | 7-amino-1-cyclopropyl-3-ethyl-2,4-dioxopyrido[2,3-d]pyrimidine-6-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
A-484954 is a highly selective inhibitor of eukaryotic elongation factor-2 kinase (eEF2K) with an IC₅₀ value of 0.28 μM in an enzymatic assay.1] |
| ln Vitro |
In an enzymatic experiment, A-484954, a highly selective eEF2K inhibitor, has an IC50 value of 280 nM for eEF2K at concentrations that are active against a range of serine/threonine and tyrosine inhibitors. In enzymatic experiments, the IC50 value of A-484954 rises as ATP concentrations rise, although calmodulin concentrations do not change this relationship [1]. A-484954 inhibited eEF2 phosphorylation (peEF2) in multiple cancer cell lines (H1299, PC3, HeLa, H460, C6) under serum-free and nutrient deprivation (HBSS) conditions, as expected for an eEF2K inhibitor. [1] In a proliferation assay using PC3 cells, A-484954 had little inhibitory effect on cell growth in the presence of serum. Under serum-free conditions, it achieved only about 40% inhibition at 100 μM, while concentrations that effectively inhibited eEF2 phosphorylation (e.g., 10-100 μM) did not result in significant growth inhibition. [1] |
| ln Vivo | NA precontraction in the E(+) and E(-) aorta and mesenteric arteries is caused by A484954. A-484954-induced mesenteric artery relaxation can be partially blocked by treatment with L-NAME in conjunction with methacin or cimetidine [2]. Long-term A-484954 administration prevents MCT-induced elevation of PA pressure. It was demonstrated that A-484954 inhibited MCT-induced PA fibrosis and hypertrophy, but not endothelial regulated or unregulated damage. Furthermore, A-484954 prevents ROS production, matrix metallochrome-2 activation, and MCT-induced NADPH oxidase-1 expression [3]. |
| Enzyme Assay |
The eEF2K enzymatic assay measured the incorporation of radiolabeled phosphate from [γ-³³P]ATP into a biotinylated myelin basic protein substrate. The reaction mixture (30 μL total volume) contained 20 mM HEPES (pH 7.4), 10 mM MgCl₂, 1 mM CaCl₂, 100 μM sodium orthovanadate, 1 mM DTT, 0.0075% Triton X-100, 10 nM calmodulin, 1 μM biotinylated myelin basic protein, 2 nM GST-eEF2K, and an ATP mixture (5 μM ATP with 10 μCi/mL of [γ-³³P]ATP). The reaction proceeded at room temperature for 45 minutes and was terminated by adding a quench buffer containing EDTA and sodium chloride. The mixture was then transferred to a streptavidin-coated plate, incubated, washed, and the bound radioactivity was measured in a scintillation counter to determine enzyme activity. [1] High-throughput screening (HTS) for eEF2K inhibitors was performed using a described format. Confirmed hits like A-484954 had their IC₅₀ values determined in a 384-well enzymatic assay from powder stocks, using nonlinear regression analysis from 6-point dose-response curves. [1] |
| Cell Assay |
For Western blot analysis of eEF2 phosphorylation, cells (e.g., H1299, PC3, HeLa, H460, C6) were seeded at 100,000 cells/well in 24-well plates. The next day, cells were treated with A-484954 at indicated concentrations (e.g., 1, 3, 10, 30, 100 μM) for 6 hours under defined conditions (e.g., serum-free medium or HBSS). Cells were then rinsed with PBS and harvested in a sample buffer containing reducing agent, protease inhibitors, and phosphatase inhibitors. Cell lysates were resolved by SDS-PAGE (4-12% gel), transferred to a PVDF membrane, and blocked. Membranes were probed overnight at 4°C with primary antibodies against phospho-eEF2 (Thr56), total eEF2, and β-actin. Detection was performed using fluorescently labeled secondary antibodies and an infrared imaging system. [1] For cell proliferation assay (CyQuant NF), cells (e.g., PC3) were seeded at 2000 cells/well in 96-well plates and treated with A-484954 for 3 days. Cell proliferation was measured using a fluorescent DNA-binding dye according to the manufacturer's protocol, and fluorescence was quantified. [1] |
| References |
[1]. 1-Benzyl-3-cetyl-2-methylimidazolium iodide (NH125) induces phosphorylation of eukaryotic elongation factor-2 (eEF2): a cautionary note on the anticancer mechanism of an eEF2 kinase inhibitor. J Biol Chem. 2011 Dec 23;286(51):43951-8. [2]. Mechanisms underlying the relaxation by A484954, a eukaryotic elongation factor 2 kinase inhibitor, in rat isolated mesenteric artery. J Pharmacol Sci. 2018 May;137(1):86-92. [3]. Eukaryotic elongation factor 2 kinase mediates monocrotaline-induced pulmonary arterial hypertension via reactive oxygen species-dependent vascular remodeling. Am J Physiol Heart Circ Physiol. 2015 May 15;308(10):H1298-305. |
| Additional Infomation |
A-484954 was identified as a highly selective small molecule inhibitor of eEF2K from a chemical library screen. It was used in this study as a pharmacological tool to demonstrate that direct inhibition of eEF2K leads to reduced phosphorylation of its substrate eEF2 in cells, but does not significantly inhibit cancer cell proliferation, contrasting with the effects of another compound, NH125. The study concluded that eEF2K inhibition itself is not an efficacious anti-proliferation strategy in the tested cancer cell lines. [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~25 mg/mL (~86.42 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.64 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2 mg/mL (6.91 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.4567 mL | 17.2837 mL | 34.5674 mL | |
| 5 mM | 0.6913 mL | 3.4567 mL | 6.9135 mL | |
| 10 mM | 0.3457 mL | 1.7284 mL | 3.4567 mL |