Physicochemical Properties
| Molecular Formula | C6H6N4O2 |
| Molecular Weight | 166.13744 |
| Exact Mass | 166.049 |
| CAS # | 552-62-5 |
| Related CAS # | 7-Methylxanthine-2,4,5,6-13C4, 1,3-15N2 (with variable 15N labeling at N9) |
| PubChem CID | 68374 |
| Appearance | White to light brown solid powder |
| Density | 1.8±0.1 g/cm3 |
| Melting Point | 300ºC |
| Index of Refraction | 1.827 |
| LogP | -0.89 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 12 |
| Complexity | 242 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | PFWLFWPASULGAN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H6N4O2/c1-10-2-7-4-3(10)5(11)9-6(12)8-4/h2H,1H3,(H2,8,9,11,12) |
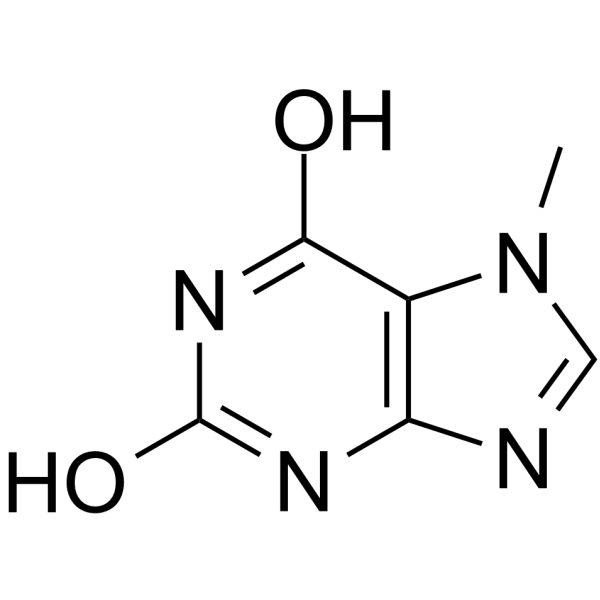
| Chemical Name | 7-methyl-3H-purine-2,6-dione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | ’- Peroxisome Proliferator-Activated Receptor α (PPARα): EC50 = 10.3 μM (determined by PPARα transactivation assay in HEK293T cells) [2] |
| ln Vitro |
1. Insecticidal activity against Culex pipiens pallens larvae: Atractylodin was tested via larval immersion method. After 24 h exposure, its median lethal concentration (LC₅₀) against Culex pipiens pallens larvae was 12.5 μg/mL; mortality reached 90% at 25 μg/mL. [1] 2. Repellent activity against Periplaneta americana adults: Filter paper discs impregnated with Atractylodin (100 μg/cm²) were used. At 2 h post-treatment, repellency rate was 80%; at 4 h, it decreased to 65%. [1] 3. Anti-inflammatory activity in LPS-stimulated RAW264.7 macrophages: Atractylodin (10, 20, 40 μM) was pre-incubated with cells for 1 h before LPS (1 μg/mL) stimulation. After 24 h, nitric oxide (NO) production was inhibited by 23.5%, 45.2%, 68.7% (vs. LPS group); 40 μM Atractylodin reduced TNF-α secretion to 65.3% and IL-6 to 58.1% of the LPS group (detected by ELISA). [2] 4. PPARα activation in HEK293T cells: Transfected HEK293T cells (with PPARα plasmid and PPRE-luciferase plasmid) were treated with Atractylodin (0.1–100 μM). Relative luciferase activity (firefly/Renilla) increased dose-dependently, with EC50 = 10.3 μM. [2] |
| ln Vivo | - Anti-colitis activity in DSS-induced mouse model: Male C57BL/6 mice (6–8 weeks old, n=6/group) were divided into DSS Model (DSS), Atractylodin 10 mg/kg (A10), Atractylodin 20 mg/kg (A20), and Normal Control (NC) groups. Colitis was induced by 3% DSS in drinking water for 7 days; Atractylodin (dissolved in 0.5% CMC-Na) was orally administered once daily for 7 days. Results: (1) Body weight: A10/A20 recovered to 91.5%/95.3% of initial weight (vs. 82.1% in DSS); (2) DAI score: 4.2/2.5 (A10/A20) vs. 6.8 (DSS); (3) Colon length: 6.2/6.8 cm (A10/A20) vs. 4.5 cm (DSS); (4) Histological score: 5.1/3.2 (A10/A20) vs. 8.5 (DSS); (5) Colonic TNF-α/IL-6: 45.1%/40.2% (A20 vs. DSS); (6) Colonic ACOX1/CPT1A mRNA: 2.1/1.8-fold higher (A20 vs. DSS, detected by qPCR). [2] |
| Enzyme Assay |
- PPARα transactivation assay: 1. HEK293T cells (5×10⁴ cells/well) were seeded in 96-well plates and cultured overnight (37°C, 5% CO₂). 2. Cells were transfected with 0.1 μg PPARα plasmid, 0.1 μg PPRE-luciferase plasmid, and 0.01 μg Renilla plasmid. 3. 24 h post-transfection, medium was replaced with fresh medium containing Atractylodin (0.1, 1, 10, 30, 100 μM) or fenofibrate (10 μM, positive control). 4. 24 h post-drug treatment, cells were lysed; luciferase activity was measured via dual-luciferase system. 5. Relative activity (firefly/Renilla) was calculated; EC50 was determined from dose-response curve. [2] |
| Cell Assay |
1. RAW264.7 macrophage NO detection assay: 1. Cells (5×10⁴ cells/well) were seeded in 96-well plates and cultured overnight. 2. Atractylodin (10, 20, 40 μM) was added for 1 h pre-incubation, followed by LPS (1 μg/mL) stimulation for 24 h. 3. 100 μL supernatant was mixed with 100 μL Griess reagent; absorbance at 540 nm was measured to calculate NO concentration. [2] 2. RAW264.7 macrophage cytokine detection assay: 1. Cells were treated as in NO assay. 2. 24 h post-LPS stimulation, supernatant was centrifuged (1000×g, 5 min); TNF-α/IL-6 concentrations were measured via ELISA. [2] |
| Animal Protocol |
- DSS-induced mouse colitis experiment: 1. Mice (20–22 g) were grouped into NC, DSS, A10 (10 mg/kg Atractylodin), A20 (20 mg/kg Atractylodin) (n=6/group). 2. DSS/A10/A20 groups received 3% DSS in drinking water for 7 days; NC received normal water. 3. Atractylodin was dissolved in 0.5% CMC-Na, orally administered once daily for 7 days (10 mL/kg body weight); NC/DSS received 0.5% CMC-Na. 4. On day 8, mice were sacrificed; colon length was measured. Colon tissue was fixed in 4% paraformaldehyde (histology) or stored at -80°C (RNA/cytokine detection). 5. Indicators: daily body weight, DAI, colon length, histological score, colonic TNF-α/IL-6, PPARα target gene expression. [2] |
| ADME/Pharmacokinetics |
Metabolism / Metabolites 7-Methylxanthine is a known human metabolite of theobromine. |
| References |
[1]. Determination of urinary 13C-caffeine metabolites by liquid chromatography-mass spectrometry: the use of metabolic ratios to assess CYP1A2 activity. J Pharm Biomed Anal. 2004 Feb 4;34(2):379-89. |
| Additional Infomation |
7-methylxanthine is an oxopurine that is xanthine in which the hydrogen attached to the nitrogen at position 7 is replaced by a methyl group. It is an intermediate metabolite in the synthesis of caffeine. It has a role as a plant metabolite, a human xenobiotic metabolite and a mouse metabolite. It is an oxopurine and a purine alkaloid. It is functionally related to a 7H-xanthine. 7-Methylxanthine has been reported in Corbicula sandai, Corbicula japonica, and other organisms with data available. - Atractylodin is a polyacetylene compound isolated from dried rhizomes of Atractylodes lancea (Thunb.) DC. [1][2] - Literature [1] suggested Atractylodin may exert insecticidal effects via disrupting insect cuticle or nervous system. [1] - Atractylodin ameliorates colitis by activating PPARα, up-regulating ACOX1/CPT1A, and down-regulating NF-κB-mediated pro-inflammatory pathway. [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~4 mg/mL (~24.08 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.0190 mL | 30.0951 mL | 60.1902 mL | |
| 5 mM | 1.2038 mL | 6.0190 mL | 12.0380 mL | |
| 10 mM | 0.6019 mL | 3.0095 mL | 6.0190 mL |