Physicochemical Properties
| Molecular Formula | C25H16O9 |
| Molecular Weight | 460.39 |
| Exact Mass | 460.079 |
| Elemental Analysis | C, 65.22; H, 3.50; O, 31.28 |
| CAS # | 3348-03-6 |
| Related CAS # | 5-CFDA;79955-27-4;5(6)-CFDA;124387-19-5 |
| PubChem CID | 151095 |
| Appearance | White to off-white solid powder |
| Density | 1.6±0.1 g/cm3 |
| Boiling Point | 701.6±60.0 °C at 760 mmHg |
| Melting Point | 152–153°C |
| Flash Point | 245.1±26.4 °C |
| Vapour Pressure | 0.0±2.3 mmHg at 25°C |
| Index of Refraction | 1.702 |
| LogP | 2.45 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 34 |
| Complexity | 828 |
| Defined Atom Stereocenter Count | 0 |
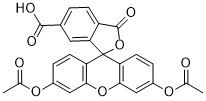
| SMILES | CC(OC1=CC=C(C2(O3)C4=CC(C(O)=O)=CC=C4C3=O)C(OC5=C2C=CC(OC(C)=O)=C5)=C1)=O |
| InChi Key | QMOGCCYGOPYYNT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H16O9/c1-12(26)31-15-4-7-18-21(10-15)33-22-11-16(32-13(2)27)5-8-19(22)25(18)20-9-14(23(28)29)3-6-17(20)24(30)34-25/h3-11H,1-2H3,(H,28,29) |
| Chemical Name | 3',6'-diacetyloxy-1-oxospiro[2-benzofuran-3,9'-xanthene]-5-carboxylic acid |
| Synonyms | CFDA-6; CFDA 6; CFDA6; 6-Carboxyfluorescein diacetate; 3348-03-6; 6-Cfda; 3',6'-Diacetoxy-3-oxo-3H-spiro[isobenzofuran-1,9'-xanthene]-6-carboxylic acid; 6-carboxyfluoresceine diacetate; 3',6'-diacetyloxy-1-oxospiro[2-benzofuran-3,9'-xanthene]-5-carboxylic acid; DTXSID80187121; MFCD00036871; 6-CFDA; 6 CFDA; 6CFDA |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluorescent Dye |
| ln Vitro |
Preparation of 6-CFDA Working Solution 1. Stock Solution Preparation • Dissolve 6-CFDA in DMSO to prepare a 10 mM stock solution. Example: Dissolve 1 mg 6-CFDA in 0.2172 mL DMSO. Note: Aliquot and store the stock solution at -20°C or -80°C, protected from light. 2. Working Solution Preparation • Dilute the stock solution with pre-warmed serum-free cell culture medium or PBS to prepare a 1-10 μM 6-CFDA working solution. Note: Adjust the working concentration as needed and prepare fresh before use. ________________________________________ Cell Staining Protocol 1. Cell Preparation • Suspension cells: Centrifuge to pellet cells, wash twice with PBS (5 min each). • Adherent cells: Remove medium, detach cells with trypsin. After centrifugation, discard supernatant and wash twice with PBS (5 min each). 2. Staining • Add 1 mL of 6-CFDA working solution and incubate at room temperature for 15 min. 3. Washing • Centrifuge at 400 × g, 4°C for 3-4 min, discard supernatant. • Wash cells twice with PBS (5 min each). 4. Resuspension & Detection • Resuspend cells in 1 mL serum-free medium or PBS. • Analyze using fluorescence microscopy or flow cytometry. ________________________________________ Precautions 1. Storage: Aliquot 6-CFDA stock solution and store at -20°C (1 month) or -80°C (6 months). Avoid repeated freeze-thaw cycles. Protect from light. 2. Concentration: Optimize the working solution concentration based on experimental conditions. 3. Use: For research purposes only. Not for clinical diagnosis, therapy, food, or drug use. 4. Safety: Wear a lab coat and disposable gloves during handling. |
| References |
[1]. A novel nonradioactive CFDA assay to monitor the cellular immune response in myeloid leukemia. Immunobiology. 2013 Apr;218(4):548-53. [2]. Assessment of fluorescein-based fluorescent dyes for tracing Neotyphodium endophytes in planta. Mycologia. 2013 Jan-Feb;105(1):221-9. [3]. Bone marrow-derived endothelial progenitor cells are involved in aneurysm repair in rabbits. J Clin Neurosci. 2012 Sep;19(9):1283-6. |
| Additional Infomation |
Endothelial progenitor cells (EPC) are believed to be involved in aneurysmal repair and remodeling. The aim of this study was to test this hypothesis and, if true, explore how EPC contribute to aneurysm repair in a rabbit model of elastase-induced carotid aneurysm. Rabbits were divided randomly into an in situ carotid EPC transfusion group (ISCT group, n=5), and an intravenous EPC transfusion group (IVT group, n=5). Autologous EPC were double-labeled with Hoechst 33342 and 5,6-carboxyfluorescein diacetate succinimidyl ester before injection into the animals in either the carotid artery (ISCT group) or marginal ear veins (IVT group). Three weeks later, labeled cells in the aneurysms were observed with respect to location, adhesion, and growth to detect signs of aneurysm repair. Labeled EPC were detected within the neointima in all five aneurysms in the ISCT group and in three of the five aneurysms in the IVT group, but there was no endothelial growth in the aneurysmal neointima in either group. These results show that bone marrow-derived EPC are involved in the process of aneurysm repair in this rabbit model. [1] Fluorescent dyes were assessed for their ability to stain viable hyphae of the fungi Neotyphodium lolii and N. coenophialum, symbiotic endophytes of the Pooideae grasses Lolium perenne and Festuca arundinacea, respectively. The fluorescein-based fluorophores; fluorescein diacetate (FDA), 5(6)-carboxy-fluorescein diacetate (CFDA), 5-chloromethylfluorescein diacetate (CMFDA) and the chitin-binding stain, Calcofluor while M2R, were assessed for staining of endophyte hyphae in vitro from axenic fungal cultures and in planta, including epidermal leaf sheath peels, nodes, ovaries, embryos and meristems. CMFDA produced the greatest intensity of staining of fungal hyphae and gave excellent contrast in planta compared to the plant cells. Compared to the other dyes, CMFDA was also the least affected by photo bleaching and continued to fluoresce up to 2 h after initial excitation. None of the fluorescent dyes stained fungal hyphae in seed. [2] Endothelial progenitor cells (EPC) are believed to be involved in aneurysmal repair and remodeling. The aim of this study was to test this hypothesis and, if true, explore how EPC contribute to aneurysm repair in a rabbit model of elastase-induced carotid aneurysm. Rabbits were divided randomly into an in situ carotid EPC transfusion group (ISCT group, n=5), and an intravenous EPC transfusion group (IVT group, n=5). Autologous EPC were double-labeled with Hoechst 33342 and 5,6-carboxyfluorescein diacetate succinimidyl ester before injection into the animals in either the carotid artery (ISCT group) or marginal ear veins (IVT group). Three weeks later, labeled cells in the aneurysms were observed with respect to location, adhesion, and growth to detect signs of aneurysm repair. Labeled EPC were detected within the neointima in all five aneurysms in the ISCT group and in three of the five aneurysms in the IVT group, but there was no endothelial growth in the aneurysmal neointima in either group. These results show that bone marrow-derived EPC are involved in the process of aneurysm repair in this rabbit model. [3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~217.21 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.43 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.43 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.43 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1721 mL | 10.8604 mL | 21.7207 mL | |
| 5 mM | 0.4344 mL | 2.1721 mL | 4.3441 mL | |
| 10 mM | 0.2172 mL | 1.0860 mL | 2.1721 mL |