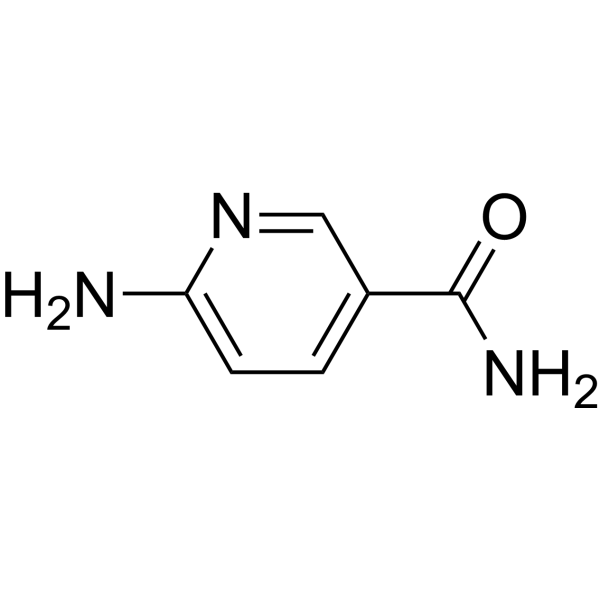
Physicochemical Properties
| Molecular Formula | C6H7N3O |
| Molecular Weight | 137.14 |
| Exact Mass | 137.058 |
| CAS # | 329-89-5 |
| PubChem CID | 9500 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 381.6±27.0 °C at 760 mmHg |
| Melting Point | 245-248 °C(lit.) |
| Flash Point | 184.6±23.7 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.644 |
| LogP | 0.41 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 10 |
| Complexity | 137 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | ZLWYEPMDOUQDBW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H7N3O/c7-5-2-1-4(3-9-5)6(8)10/h1-3H,(H2,7,9)(H2,8,10) |
| Chemical Name | 6-aminopyridine-3-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | 6-Aminonicotinamide (100 nM; 7 days) dramatically lowers AR+ in CRPC-derived C4-2 and 22Rv1 cell models, human AR+, and hormone-sensitive prostate cancer cell lines LNCaP and LAPC4. Basal and R1881-mediated ROS levels are increased by 6-aminonicotinamide (100 nM ± 10 nM R1881) for three days, suggesting that 6-aminonicotinamide is obstructing the cell's antioxidant defenses. In C4-2 cells, 6-aminonicotinamide can also raise ROS levels [3]. |
| ln Vivo | 6-Aminonicotinamide (20 mg/kg; intraperitoneally; stimulation time; days 1, 10, or 11 and 21) by itself caused a little but notable halt in tumor growth (4.3 +/- 0.8 days). Treatment with 6-Aminonicotinamide and radiation caused a tumor growth delay of 57.0+/-3.8 days in a CD8F1 breast tumor model [4]. |
| References |
[1]. TRIM21 and PHLDA3 negatively regulate the crosstalk between the PI3K/AKT pathway and PPP metabolism. Nat Commun. 2020;11(1):1880. Published 2020 Apr 20. [2]. Influence of 6-aminonicotinamide (6AN) on Leishmania promastigotes evaluated by metabolomics: Beyond the pentose phosphate pathway. Chem Biol Interact. 2018;294:167-177. [3]. Regulation of the pentose phosphate pathway by an androgen receptor-mTOR-mediated mechanism and its role in prostate cancer cell growth. Oncogenesis. 2014;3(5):e103. Published 2014 May 26. [4]. Effect of 6-aminonicotinamide on the pentose phosphate pathway: 31P NMR and tumor growth delay studies. Magn Reson Med. 1996;36(6):887-892. |
| Additional Infomation |
6-aminonicotinamide is a white crystalline solid. (NTP, 1992) 6-aminonicotinamide is a monocarboxylic acid amide resulting from the formal condensation of the carboxy group of 6-aminonicotinic acid with ammonia. An inhibitor of the NADP(+)-dependent enzyme, 6-phosphogluconate dehydrogenase, it interferes with glycolysis, resulting in ATP depletion and synergizes with DNA-crosslinking chemotherapy drugs, such as cisplatin, in killing cancer cells. It has a role as a teratogenic agent, an antimetabolite and an EC 1.1.1.44 (NADP(+)-dependent decarboxylating phosphogluconate dehydrogenase) inhibitor. It is an aminopyridine, a primary amino compound and a monocarboxylic acid amide. It is functionally related to a 6-aminonicotinic acid. 6-Aminonicotinamide is a synthetic analogue and nicotinamide antagonist with cytotoxic properties, 6-Aminonicotinamide forms an abnormal NAD (ubiquitous metabolic pathway coenzyme) analog that inhibits NAD-dependent enzyme activity associated with ATP production. 6-Aminonicotinamide impairs cartilage formation, may have teratogenic effects, and enhances the effect of radiation and cisplatin treatment (formation of platinum-DNA complexes). (NCI04) A vitamin antagonist which has teratogenic effects. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 100 mg/mL (729.18 mM) H2O: 11.11 mg/mL (81.01 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (18.23 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (18.23 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (18.23 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 4 mg/mL (29.17 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.2918 mL | 36.4591 mL | 72.9182 mL | |
| 5 mM | 1.4584 mL | 7.2918 mL | 14.5836 mL | |
| 10 mM | 0.7292 mL | 3.6459 mL | 7.2918 mL |