Physicochemical Properties
| Molecular Formula | C₁₂H₁₅N₃O |
| Molecular Weight | 217.27 |
| Exact Mass | 217.121 |
| CAS # | 519-98-2 |
| Related CAS # | 4-Methylamino antipyrine hydrochloride;856307-27-2 |
| PubChem CID | 10618 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 324.5±45.0 °C at 760 mmHg |
| Melting Point | 50-53ºC |
| Flash Point | 150.0±28.7 °C |
| Vapour Pressure | 0.0±0.7 mmHg at 25°C |
| Index of Refraction | 1.619 |
| LogP | 0.02 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 16 |
| Complexity | 318 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | JILCEWWZTBBOFS-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H15N3O/c1-9-11(13-2)12(16)15(14(9)3)10-7-5-4-6-8-10/h4-8,13H,1-3H3 |
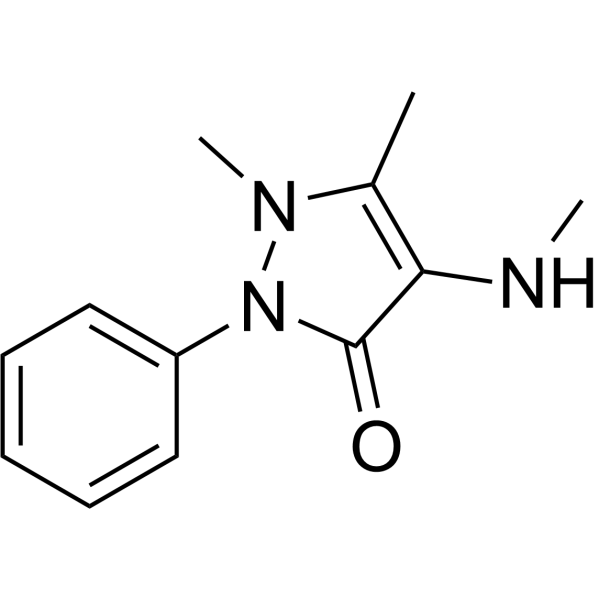
| Chemical Name | 1,5-dimethyl-4-(methylamino)-2-phenylpyrazol-3-one |
| Synonyms | 4Methylamino antipyrine; 4 Methylamino antipyrine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In an oxygenated environment and at ambient temperature, the prodrug methimizole spontaneously and non-enzymatically transforms into 4-methylaminoantipyrine. In order to produce 4-formamidoantipyrine, which is then transformed into 4-aminoantipyrine, the N-methyl side chain of 4-methylaminoantipyrine is oxidized. The most significant pyrazolone derivative in terms of pharmacology is 4-methylaminoantipyrine, which is part of the group of compounds known as metamizole when present in an aqueous solution and with oxygen [2]. |
| ln Vivo | This study looked at the pharmacokinetics of 4-methylaminoantipyrine, the drug's active metabolite, following a single intramuscular injection of metamizole in male piglets. Metamizole (100 mg/kg) was given intramuscularly into eight healthy male piglets. 4- Quantitative measurements of methylaminoantipyrine plasma concentrations can be made in 0.25 to 48 hours. The 4-methylaminoantipyrine mean maximum concentration was 47.59 mg/mL. 4-methylaminoantipyrine has an average half-life of 8.57 hours [1]. |
| References |
[1]. Pharmacokinetic investigations of the marker active metabolites 4-methylamino-antipyrine and 4-amino-antipyrine after intramuscular injection of metamizole in healthy piglets. J Vet Pharmacol Ther. 2016 Dec;39(6):616-620. [2]. Regulation of cyclooxygenase activity by metamizol. Eur J Pharmacol. 1999 Aug 13;378(3):339-47. [3]. Pyrazolones metabolites are relevant for identifying selective anaphylaxis to metamizole. Sci Rep. 2016 Mar 31;6:23845. |
| Additional Infomation |
4-(methylamino)antipyrine is a member of the class of pyrazoles that is antipyrine substituted at C-4 by a methylamino group. It is a metabolite of aminopyrine and of metamizole. It has a role as a non-narcotic analgesic, an opioid analgesic, a non-steroidal anti-inflammatory drug, an EC 1.14.99.1 (prostaglandin-endoperoxide synthase) inhibitor, a peripheral nervous system drug, an antipyretic and a drug metabolite. It is a member of pyrazoles and a secondary amino compound. It is functionally related to an antipyrine. Noramidopyrine has been reported in Euglena gracilis with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~230.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.51 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.51 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.6026 mL | 23.0128 mL | 46.0257 mL | |
| 5 mM | 0.9205 mL | 4.6026 mL | 9.2051 mL | |
| 10 mM | 0.4603 mL | 2.3013 mL | 4.6026 mL |