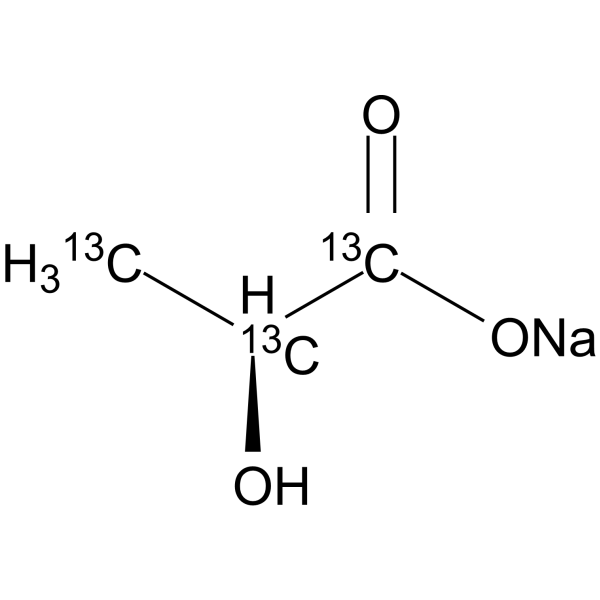
Physicochemical Properties
| Molecular Formula | 13C3H5NAO3 |
| Molecular Weight | 115.0377 |
| Exact Mass | 115.024 |
| CAS # | 201595-71-3 |
| Related CAS # | L-Lactic acid-13C3;87684-87-5 |
| PubChem CID | 23666456 |
| Appearance | Colorless to light yellow liquid |
| Melting Point | 163-165 ℃(lit.) |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 7 |
| Complexity | 63.2 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | [Na+].[13CH3][13C@@H]([13C](=O)[O-])O |
| InChi Key | NGSFWBMYFKHRBD-UHFFFAOYSA-M |
| InChi Code | InChI=1S/C3H6O3.Na/c1-2(4)3(5)6;/h2,4H,1H3,(H,5,6);/q;+1/p-1 |
| Chemical Name | sodium;2-hydroxypropanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. L-lactic acid production by Lactobacillus rhamnosus ATCC 10863. ScientificWorldJournal. 2015;2015:501029. |
| Additional Infomation |
Sodium lactate is an organic sodium salt having lactate as the counterion. It has a role as a food preservative and a food acidity regulator. It is an organic sodium salt and a lactate salt. It contains a lactate. Sodium Lactate is a sodium salt of racemic or inactive lactic acid with alkalinizing and electrolyte replenishing property. Upon metabolism, sodium lactate is converted to bicarbonate, thereby increasing plasma bicarbonate, which facilitates removal of hydrogen ion and lactate from blood stream and leads to raised blood pH. Compound Sodium Lactate Solution is a multiple electrolyte, isotonic, crystalloid solution for intravenous infusion containing sodium chloride, potassium chloride, calcium chloride dihydrate, and sodium lactate, which can restore the electrolyte balance, normalize pH, and provide water for hydration. Upon intravenous administration, the compound sodium lactate solution will replace any lost body fluids and electrolytes thereby providing hydration as well as normalizing electrolyte concentrations. In addition, conversion of sodium lactate to bicarbonate increases plasma bicarbonate levels, which facilitates the removal of hydrogen ions from the blood stream, raises blood pH and normalizes the acid-base balance. The sodium salt of racemic or inactive lactic acid. It is a hygroscopic agent used intravenously as a systemic and urinary alkalizer. See also: Sodium Cation (has active moiety) ... View More ... |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~869.26 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (21.73 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (21.73 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (21.73 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 8.6926 mL | 43.4631 mL | 86.9263 mL | |
| 5 mM | 1.7385 mL | 8.6926 mL | 17.3853 mL | |
| 10 mM | 0.8693 mL | 4.3463 mL | 8.6926 mL |