2-NBDG, an analog of 1-NBDG, is a fluorescent analogue of glucose and an indicator used for measuring glucose uptake by bacteria and live mammalian cells and in tumor biopsies. The uptake of 2-NBDG is competitively inhibited by D-glucose, but not L-glucose or sucrose, in E. coli. Evaluation of glucose uptake ability in cells plays a fundamental role in diabetes mellitus research. In this study, we describe a sensitive and non-radioactive assay for direct and rapid measuring glucose uptake in single, living cells. The assay is based on direct incubation of mammalian cells with a fluorescent d-glucose analog 2-[N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl) amino]-2-deoxy-D-glucose (2-NBDG) followed by flow cytometric detection of fluorescence produced by the cells. A series of experiments were conducted to define optimal conditions for this assay. By this technique, it was found that insulin lost its physiological effects on cells in vitro meanwhile some other anti-diabetic drugs facilitated the cell glucose uptake rates with mechanisms which likely to be different from those of insulin or those that were generally accepted of each drug. Our findings show that this technology has potential for applications in both medicine and research.
Physicochemical Properties
| Molecular Formula | C12H14N4O8 |
| Molecular Weight | 342.26200 |
| Exact Mass | 342.081 |
| Elemental Analysis | C, 42.11; H, 4.12; N, 16.37; O, 37.40 |
| CAS # | 186689-07-6 |
| Related CAS # | 2376921-70-7 (1-NBDG) |
| PubChem CID | 6711157 |
| Appearance | Typically exists as light yellow to orange solids at room temperature |
| Density | 1.750±0.06 g/cm3 |
| Boiling Point | 707.6±70.0 °C at 760 mmHg |
| Flash Point | 381.7±35.7 °C |
| Vapour Pressure | 0.0±2.4 mmHg at 25°C |
| Index of Refraction | 1.770 |
| LogP | -0.41 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 24 |
| Complexity | 449 |
| Defined Atom Stereocenter Count | 4 |
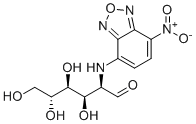
| SMILES | O=C[C@H](NC1=CC=C([N+]([O-])=O)C2=NON=C21)[C@H]([C@@H]([C@@H](CO)O)O)O |
| InChi Key | QUTFFEUUGHUPQC-ILWYWAAHSA-N |
| InChi Code | InChI=1S/C12H14N4O8/c17-3-6(11(20)12(21)8(19)4-18)13-5-1-2-7(16(22)23)10-9(5)14-24-15-10/h1-3,6,8,11-13,18-21H,4H2/t6-,8+,11+,12+/m0/s1 |
| Chemical Name | 2-deoxy-2-[(7-nitro-2,1,3-benzoxadiazol-4-yl)amino]-D-glucose |
| Synonyms | 2-NBDG; 2NBDG; 186689-07-6; JE4F4P486R; 2-Deoxy-2-((7-nitro-2,1,3-benzoxadiazol-4-yl)amino)-D-glucose; 2-Deoxy-2-[(7-nitro-2,1,3-benzoxadiazol-4-yl)amino]-D-glucose; DTXSID001348102; 2-(N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino)-2-deoxy-D-glucose; DTXCID00375967; 2 NBDG |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluorescent Dye; 2-NBDG, a glucose analog, is taken up by cells primarily via glucose transporters (e.g., GLUT family). |
| ln Vitro |
1. Preparation of working solution 1.1 Preparation of stock solution. To obtain 1 mM stock solution, dissolve 1 mg of 2-NBDG in 2.92 mL DDH2O. 1.2 Preparation of working solution: Dilute the stock solution with preheated serum-free cell culture medium or PBS to prepare 10-200 μ M's 2-NBDG working solution. Note: Please adjust the concentration of 2-NBDG working solution according to your specific needs. 2. Cell staining 2.1 Suspended cells: Centrifuge and collect cells, add PBS and wash twice, each time for 5 minutes. Adherent cells: Discard the culture medium and add trypsin to digest the cells. After centrifugation, discard the supernatant and wash twice with PBS for 5 minutes each time. 2.2 Add 1 mL of 2-NBDG working solution and incubate at room temperature for 5-60 minutes. 2.3 At 400 g, centrifuge at 4 ℃ for 3-4 minutes, discard the supernatant. 2.4 Add PBS to wash cells twice, each time for 5 minutes. 2.5 After resuspend cells with 1 mL of serum-free medium or PBS, observe under a microscope. If viability testing is performed, record the optical density (O.D.) at 540/570 nm using an ELISA enzyme-linked immunosorbent assay (ELISA) reader. Cell viability was calculated using a control ratio and plotted against the logarithmic concentration of the drug to calculate IC50. - Cellular uptake activity: 2-NBDG can be actively taken up by various mammalian cells (e.g., 3T3-L1 adipocytes, HeLa cells, HepG2 cells), and the uptake amount is associated with cellular metabolic activity. Fluorescence intensity increases with incubation time (5-60 minutes). At physiological glucose concentrations (5-25 mM), uptake is competitively inhibited by excess unlabeled glucose or glucose transport inhibitors (e.g., phlorizin), indicating that uptake is dependent on glucose transport systems[1] - Specificity verification: In glucose-free medium, cellular uptake of 2-NBDG is significantly enhanced; in the presence of high glucose (50 mM), uptake is inhibited by approximately 70%-80%, confirming glucose-specific uptake[2] - Association with metabolic status: In differentiated 3T3-L1 adipocytes, insulin treatment promotes 2-NBDG uptake (by ~2-3 fold), indicating that it can reflect insulin-regulated glucose transport activity[1] |
| ln Vivo |
Circulating breast cancer cells with increased uptake of fluorescent 2-NBDG were detected in mice bearing human breast cancer xenograft tumors by fluorescence imaging, suggesting clinical use of 2-NBDG as a tracer for fluorescence imaging of hypermetabolic circulating breast cancer cells [3].
- Cellular uptake activity: 2-NBDG can be actively taken up by various mammalian cells (e.g., 3T3-L1 adipocytes, HeLa cells, HepG2 cells), and the uptake amount is associated with cellular metabolic activity. Fluorescence intensity increases with incubation time (5-60 minutes). At physiological glucose concentrations (5-25 mM), uptake is competitively inhibited by excess unlabeled glucose or glucose transport inhibitors (e.g., phlorizin), indicating that uptake is dependent on glucose transport systems[1] - Specificity verification: In glucose-free medium, cellular uptake of 2-NBDG is significantly enhanced; in the presence of high glucose (50 mM), uptake is inhibited by approximately 70%-80%, confirming glucose-specific uptake[2] - Association with metabolic status: In differentiated 3T3-L1 adipocytes, insulin treatment promotes 2-NBDG uptake (by ~2-3 fold), indicating that it can reflect insulin-regulated glucose transport activity[1] |
| Enzyme Assay | Fluorescence Imaging of Circulating Breast Cancer Cells with Increased Uptake of Fluorescent 2-NBDG Starting at 1 week after implantation of tumor cells, mouse blood samples (100 μL/mouse) were collected by puncturing the mouse saphenous vein. The blood samples containing circulating breast cancer cells were incubated with 2-NBDG, a fluorescent glucose analogue purchased from Invitrogen, at a dose of 5 μg/100 μL blood, for 30 min in a dark incubator at 37 °C. Subsequently, circulating breast cancer cells were harvested with magnetic beads conjugated with anti-epithelial cell adhesion molecule (EpCAM) IgG in a protocol from the vendor, followed by fluorescence imaging of circulating breast cancer cells with cellular uptake of fluorescent 2-NBDG. Briefly, 1 μL of 1 % suspension of the magnetic beads was added to 100 μL of blood samples at the end of incubation with 2-NBDG, followed by incubation at 4 °C for 30 min with gentle shaking to facilitate binding of the magnetic beads to circulating breast cancer cells. Subsequently, the circulating breast cancer cells were collected from the blood by magnetic separation with a magnetic separation rack, washed 3 times with PBS, and transferred to a 96-well plate after re-suspending the circulating breast cancer cells in 100 μl PBS. Uptake of 2-NBDG by circulating breast cancer cells was examined under a fluorescent microscope equipped with a 488 nm filter (Olympus). Large cells with fluorescent signals derived from cellular uptake of fluorescent 2-NBDG were counted as hypermetabolic circulating breast cancer cells, in comparison to small size of normal mouse blood cells (lymphocytes and RBC) showing no or little fluorescence signals of 2-NBDG. Total number of hypermetabolic circulating breast cancer cells in a blood sample was obtained by visual scanning of the whole area of the well of the 96 well plate with manual cell counter. The experiments were repeated three times[3]. |
| Cell Assay |
Staining Example 1 for 2-NBDG uptake assay: 2-NBDG may be used as a fluorescent indicator for monitoring the uptake of glucose in living cells. 1. Incubate cells with 2-NBDG (50 μM; 30 min; 37℃; 5% CO2) in glucose-free DMEM. 2. Wash cells thoroughly and measure the fluorescence by microscope. Staining Example 2 for 2-NBDG uptake assay:2-NBDG may be used as a fluorescent indicator for monitoring the uptake of glucose in living cells. 1. Incubate cells with low-glucose DMEM containing 2-NBDG (150 μg/mL; 60 min; 37℃). 2. Wash cells thoroughly and measure the fluorescence by microscope. Staining Example 3 for 2-NBDG uptake assay:2-NBDG may be used as a fluorescent indicator for monitoring the uptake of glucose in living cells. 1. Incubate cells in low-glucose DMEM with 2-NBDG (150 μg/mL; 60 min; 37℃). 2. Wash cells thoroughly and measure the fluorescence by microscope. Staining Example 4 for 2-NBDG uptake assay: 2-NBDG may be used as a fluorescent indicator for monitoring the uptake of glucose in living cells. 1. Incubate cells with 2-NBDG (100 μM; 30 min). 2. Wash cells thoroughly and measure the fluorescence by microscope. - Basic uptake assay protocol: Log-phase cells are seeded in culture plates and pre-treated with glucose-free medium for 30 minutes; 2-NBDG is added at a final concentration of 10-50 μM, followed by incubation at 37°C for 5-60 minutes; after incubation, cells are washed 3 times with ice-cold PBS to remove unuptaken probe; intracellular fluorescence intensity is detected by fluorescence microscopy (excitation 465-495 nm, emission 515-555 nm) or flow cytometry[1] - Insulin regulation assay protocol: Differentiated 3T3-L1 adipocytes are starved in serum-free medium for 12 hours; pre-treated with 100 nM insulin for 30 minutes, then incubated with 20 μM 2-NBDG for 30 minutes; after washing, uptake changes are quantified by fluorescence to evaluate insulin-promoted glucose transport[1] - Other cell assay protocols: Different concentrations of 2-NBDG (e.g., 25 μM) are added to cells and incubated for a specific time (e.g., 30 minutes); fluorescence intensity distribution is analyzed by flow cytometry to compare uptake capacity among different cell lines[2] |
| Animal Protocol |
Procedures:[4] Human breast cancer cells were implanted in the mammary gland fat pad of athymic mice to establish orthotopic human breast cancer xenografts as a mouse model of circulating breast cancer cells. Near-infrared fluorescence imaging of the tumor-bearing mice injected with 2-DeoxyGlucosone 750 (2-DG 750) was conducted to assess glucose metabolism of xenograft tumors. Following incubation with fluorescent 2-NBDG, circulating breast cancer cells in the blood samples collected from the tumor-bearing mice were collected by magnetic separation, followed by fluorescence imaging for 2-NBDG uptake by circulating breast cancer cells, and correlation of the number of hypermetabolic circulating breast cancer cells with tumor size at the time when the blood samples were collected. Results: [4] Human breast cancer xenograft tumors derived from MDA-MB-231, BT474, or SKBR-3 cells were visualized on near-infrared fluorescence imaging of the tumor-bearing mice injected with 2-DG 750. Hypermetabolic circulating breast cancer cells with increased uptake of fluorescent 2-NBDG were detected in the blood samples from tumor-bearing mice and visualized by fluorescence imaging, but not in the blood samples from normal control mice. The number of hypermetabolic circulating breast cancer cells increased along with growth of xenograft tumors, with the number of hypermetabolic circulating breast cancer cells detected in the mice bearing MDA-MB231 xenografts larger than those in the mice bearing BT474 or SKBR-3 xenograft tumors. Mouse xenograft model experiment: Human breast cancer cells (MDA-MB-231) are subcutaneously inoculated into the axilla of female nude mice (6-8 weeks old) to establish tumor models; when tumor volume reaches 100-200 mm³, 2-NBDG is injected via tail vein (dose: 100-200 μL, concentration 5 mM); at 15-30 minutes after injection, peripheral blood is collected, mononuclear cells are isolated by density gradient centrifugation, and 2-NBDG-positive circulating tumor cells are detected by fluorescent flow cytometry (excitation 488 nm, emission 530 nm)[3] |
| ADME/Pharmacokinetics |
Detailed ADME parameters (absorption, distribution, metabolism, excretion) of 2-NBDG are not described in the literature, except that fluorescent signals can be detected in target cells/tissues at 15-30 minutes after in vivo injection[3] |
| Toxicity/Toxicokinetics | No significant toxicity of 2-NBDG is reported. In in vitro cell experiments (concentration ≤50 μM) and in vivo animal experiments (injection dose ≤200 μL of 5 mM solution), no increased cell death or abnormal animal behavior is observed |
| References |
[1]. A real-time method of imaging glucose uptake in single, living mammalian cells. Nat Protoc. 2007;2(3):753-62. [2]. 2-NBDG as a fluorescent indicator for direct glucose uptake measurement. J Biochem Biophys Methods. 2005 Sep 30;64(3):207-15. [3]. 2-NBDG fluorescence imaging of hypermetabolic circulating tumor cells in mouse xenograft model of breast cancer. J Fluoresc. 2013 Jan;23(1):213-20. |
| Additional Infomation |
This protocol details a method for monitoring glucose uptake into single, living mammalian cells using a fluorescent D-glucose derivative, 2-[N-(7-nitrobenz-2-oxa-1,3-diazol-4-yl)amino]-2-deoxy-D-glucose (2-NBDG), as a tracer. The specifically designed chamber and superfusion system for evaluating 2-NBDG uptake into cells in real time can be combined with other fluorescent methods such as Ca2+ imaging and the subsequent immunofluorescent classification of cells exhibiting divergent 2-NBDG uptake. The whole protocol, including immunocytochemistry, can be completed within 2 d (except for cell culture). The procedure for 2-NBDG synthesis is also presented.[1]
- Mechanism of action: 2-NBDG is a fluorescently labeled D-glucose analog that retains the pyranose ring of glucose. It can be taken up by cells via glucose transporters but cannot be further metabolized due to the replacement of the 6-hydroxyl group with a fluorescent moiety, thus accumulating in cells and emitting fluorescence, which allows real-time monitoring of glucose uptake activity[1] [2] - Application advantages: Compared with radioactive glucose probes (e.g., FDG), 2-NBDG does not require radioactive detection equipment and can be detected by conventional fluorescence microscopy or flow cytometry, making it suitable for real-time imaging at the single-cell level[1][2] |
Solubility Data
| Solubility (In Vitro) | H2O : ~5 mg/mL (~14.61 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 3.33 mg/mL (9.73 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9218 mL | 14.6088 mL | 29.2176 mL | |
| 5 mM | 0.5844 mL | 2.9218 mL | 5.8435 mL | |
| 10 mM | 0.2922 mL | 1.4609 mL | 2.9218 mL |