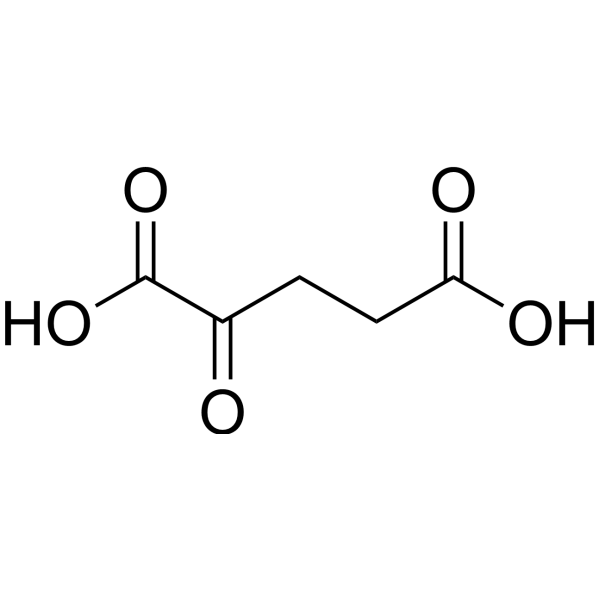
Physicochemical Properties
| Molecular Formula | C5H6O5 |
| Molecular Weight | 146.09814 |
| Exact Mass | 146.021 |
| CAS # | 328-50-7 |
| Related CAS # | 2-Ketoglutaric acid Sodium;22202-68-2;2-Ketoglutaric acid-13C5;161096-83-9;2-Ketoglutaric acid-13C;108395-15-9;Calcium 2-oxoglutarate;71686-01-6;2-Ketoglutaric acid-d4;1381759-60-9;2-Ketoglutaric acid-d6;1173021-86-7 |
| PubChem CID | 51 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 345.6±25.0 °C at 760 mmHg |
| Melting Point | 113-115 °C |
| Flash Point | 177.0±19.7 °C |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.494 |
| LogP | -1.43 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 10 |
| Complexity | 171 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | OC(=O)CCC(=O)C(O)=O |
| InChi Key | KPGXRSRHYNQIFN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C5H6O5/c6-3(5(9)10)1-2-4(7)8/h1-2H2,(H,7,8)(H,9,10) |
| Chemical Name | 2-oxopentanedioic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
2-Ketoglutaric acid targets 2-oxoglutarate-dependent dioxygenases (serves as a co-substrate) [1] 2-Ketoglutaric acid targets tyrosinase (Ki = 1.86 mM) [2] |
| ln Vitro |
In addition to its physiological roles, 2-ketoglutarate (also known as alpha-ketoglutarate) also protects against lipid peroxidation, reduces ammonia levels produced in the lungs, and provides neuroprotection against cyanide poisoning [1]. The synthesis of nucleotides and amino acids requires 2-oxoglutarate as a precursor [2]. - As a co-substrate, 2-Ketoglutaric acid activates 2-oxoglutarate-dependent dioxygenases, which are involved in diverse biological processes including collagen biosynthesis, histone/DNA demethylation, and proline hydroxylation [1] - 2-Ketoglutaric acid exhibited dose-dependent inhibitory activity against tyrosinase; at 5 mM, it inhibited tyrosinase activity by 68%, and the inhibition followed a non-competitive kinetic mechanism [2] - Molecular dynamics simulation showed that 2-Ketoglutaric acid bound to the active site of tyrosinase, inducing conformational changes in the enzyme’s catalytic pocket and reducing its affinity for the natural substrate L-tyrosine [2] - The inhibitory effect of 2-Ketoglutaric acid on tyrosinase was reversible, with enzyme activity recovering by 85% after dialysis to remove the compound [2] |
| Enzyme Assay |
- 2-Oxoglutarate-dependent dioxygenase activity assay: The reaction system contained recombinant 2-oxoglutarate-dependent dioxygenase, substrate (e.g., proline-containing peptide, methylated histone), 2-Ketoglutaric acid (0.1-5 mM), Fe²⁺, and ascorbate; the reaction was incubated at 37°C for 30-60 minutes, and the product (e.g., hydroxylated peptide, demethylated histone) was detected by chromatographic or mass spectrometric methods to evaluate enzyme activity [1] - Tyrosinase activity assay: The assay was performed in a buffer system with L-dopa as the substrate; serial concentrations of 2-Ketoglutaric acid (0.1-10 mM) were added to the mixture of tyrosinase and L-dopa, and the oxidation of L-dopa to dopachrome was monitored spectrophotometrically at 475 nm for 10 minutes; kinetic parameters (Km, Vmax, Ki) were calculated by nonlinear regression analysis [2] - Tyrosinase conformational analysis: Molecular dynamics simulation was conducted using tyrosinase crystal structure; 2-Ketoglutaric acid was docked into the enzyme’s active site, and the complex was subjected to simulation for 100 ns to analyze changes in protein backbone flexibility, hydrogen bonding, and active pocket volume [2] |
| References |
[1]. Huergo LF, et al. The Emergence of 2-Oxoglutarate as a Master Regulator Metabolite. Microbiol Mol Biol Rev. 2015 Dec;79(4):419-35. [2]. Gou L, et al. The effect of alpha-ketoglutaric acid on tyrosinase activity and conformation: Kinetics and molecular dynamics simulation study. Int J Biol Macromol. 2017 Dec;105(Pt 3):1654-1662. |
| Additional Infomation |
2-oxoglutaric acid is an oxo dicarboxylic acid that consists of glutaric acid bearing an oxo substituent at position 2. It is an intermediate metabolite in Krebs cycle. It has a role as a fundamental metabolite. It is functionally related to a glutaric acid. It is a conjugate acid of a 2-oxoglutarate(1-). Oxogluric acid (α-Ketoglutarate) is not approved for any indication in the world but is an investigational drug in the United States. In the United States a phase I clinical trial is investigating whether oxogluric acid precursors found in nutritional supplements can benefit patients with the metabolic disorder propionic acidemia. Oxogluric acid is sold as a dietary supplement to athletes to improve their performance by helping to remove excess ammonia, but it is not officially approved for this indication and only experimental studies have shown a reduction in ammonia by oxogluric acid in hemodialysis patients. Physiologically, oxogluric acid acts in the Krebs cycle as an intermediate, is involved in transamination reactions during the metabolism of amino acids, forms glutamic acid by combining with ammonia, and reduces nitrogen by combining with it as well. Several experimental studies have also shown that administration of oxogluric acid helped attenuate the decreased synthesis of muscle protein that is often seen post-surgery. Oxoglutaric acid is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). alpha-Ketoglutarate is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). 2-Oxoglutaric acid has been reported in Humulus lupulus, Drosophila melanogaster, and other organisms with data available. A family of compounds containing an oxo group with the general structure of 1,5-pentanedioic acid. (From Lehninger, Principles of Biochemistry, 1982, p442) Drug Indication α-α-Ketoglutarate is not approved for any indication in the world but is an investigational drug in the United States. The potential indications for α-Ketoglutarate are in patients with the metabolic disorder propionic acidemia and in trauma patients with muscle loss. Mechanism of Action The exact mechanisms of action for α-Ketoglutarate have not yet been elucidated. Some of α-Ketoglutarate’s actions include acting in the Krebs cycle as an intermediate, transamination reactions during the metabolism of amino acids, forming glutamic acid by combining with ammonia, and reducing nitrogen by combining with it as well. Concerning α-Ketoglutarate’s actions with ammonia, it is proposed that α-Ketoglutarate can help patients with propionic academia who have high levels of ammonia and low levels of glutamine/glutamate in their blood. Because endogenous glutamate/glutamine is produced from α-Ketoglutarate, propionic acidemia patients have impaired production of α-Ketoglutarate and supplementation of α-Ketoglutarate should improve the condition of these patients. Several other experimental studies have also shown that administration of α-Ketoglutarate in parenteral nutrition given to post-operative patients helped attenuate the decreased synthesis of muscle protein that is often seen after a surgery. This decreased muscle synthesis is speculated to be due to too low α-Ketoglutarate levels. Pharmacodynamics All of the physiological roles of alpha-ketoglutarate have not been determined. What is known is that alpha-keotglutarate is involved in the Krebs cycle, transamination reactions, and promotes muscle synthesis. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~250 mg/mL (~1711.16 mM) H2O : ~50 mg/mL (~342.23 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (14.24 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (14.24 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (14.24 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 100 mg/mL (684.46 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.8446 mL | 34.2231 mL | 68.4463 mL | |
| 5 mM | 1.3689 mL | 6.8446 mL | 13.6893 mL | |
| 10 mM | 0.6845 mL | 3.4223 mL | 6.8446 mL |