Physicochemical Properties
| Molecular Formula | C8H5O3F13S |
| Molecular Weight | 428.1677 |
| Exact Mass | 427.97518 |
| CAS # | 27619-97-2 |
| PubChem CID | 119688 |
| Appearance | Typically exists as solids at room temperature |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 221.2ºC at 760 mmHg |
| Flash Point | 87.6ºC |
| Vapour Pressure | 0.109mmHg at 25°C |
| Index of Refraction | 1.33 |
| LogP | 3.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 25 |
| Complexity | 585 |
| Defined Atom Stereocenter Count | 0 |
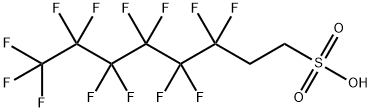
| SMILES | C(CS(=O)(=O)O)C(C(C(C(C(C(F)(F)F)(F)F)(F)F)(F)F)(F)F)(F)F |
| InChi Key | VIONGDJUYAYOPU-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H5F13O3S/c9-3(10,1-2-25(22,23)24)4(11,12)5(13,14)6(15,16)7(17,18)8(19,20)21/h1-2H2,(H,22,23,24) |
| Chemical Name | 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluorooctane-1-sulfonic acid |
| Synonyms | 1-Octanesulfonic acid, 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluoro-; 6:2 FTSA; 27619-97-2; 1h,1h,2h,2h-perfluorooctanesulfonic acid; 3,3,4,4,5,5,6,6,7,7,8,8,8-tridecafluorooctane-1-sulfonic acid; 6:2 Fluorotelomer sulfonic acid; THPFOS; 6:2 FTS; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Contaminant |
| ln Vitro | Per- and polyfluoroalkyl substances (PFASs) contamination in the Bohai Sea and its surrounding rivers has attracted considerable attention in recent years. However, few studies have been conducted regarding the distribution of PFASs in multiple environmental media and their distributions between the suspended particles and dissolved phases. In this study, surface water, surface sediment, and air samples were collected at the Bohai Sea to investigate the concentration and distribution of 39 targeted PFASs. Moreover, river water samples from 35 river estuaries were collected to estimate PFAS discharge fluxes to the Bohai Sea. The results showed that total ionic compound (Σi-PFASs) concentrations ranged from 19.3 to 967 ng/L (mean 125 ± 152 ng/L) in the water and 0.70-4.13 ng/g dw (1.78 ± 0.76 ng/g) in surface sediment of the Bohai Sea, respectively. In the estuaries, Σi-PFAS concentrations were ranged from 10.5 to 13500 ng/L (882 ± 2410 ng/L). In the air, ΣPFAS (Σi-PFASs + Σn-PFASs) concentrations ranged from 199 to 678 pg/m3 (462 ± 166 pg/m3). Perfluorooctanoic acid (PFOA) was the predominant compound in the seawater, sediment, and river water; in the air, 8:2 fluorotelomer alcohol was predominant. Xiaoqing River discharged the largest Σi-PFAS flux to the Bohai Sea, which was estimated as 12,100 kg/y. Some alternatives, i.e., 6:2 fluorotelomer sulfonate acid (6:2 FTSA), hexafluoropropylene oxide dimer acid (HFPO-DA), and chlorinated 6:2 polyfluorinated ether sulfonic acid (Cl-6:2 PFESA), showed higher levels than or comparable concentrations to those of the C8 legacy PFASs in some sampling sites. The particle-derived distribution coefficient in seawater was higher than that in the river water. Using high resolution mass spectrometry, 29 nontarget emerging PFASs were found in 3 river water and 3 seawater samples. Further studies should be conducted to clarify the sources and ecotoxicological effects of these emerging PFASs in the Bohai Sea area.[1] |
| ln Vivo | Per- and polyfluoroalkyl substances (PFAS) were analysed in a high number of terrestrial samples of soil, earthworm, bird eggs and liver from red fox and brown rat in an urban area in Norway from 2013 to 2020. PFOS and the long chain PFCAs were the most dominating compounds in all samples, proving their ubiquitous distribution. Other less studied compounds such as 6:2 FTS were first and foremost detected in earthworm. 8:2 FTS was found in many samples of fieldfare egg, sparrowhawk egg and earthworm, where the eggs had highest concentrations. Highest concentrations for both 6:2 FTS and 8:2 FTS were detected at present and former industry areas. FOSA was detected in many samples of the species with highest concentrations in red fox liver and brown rat liver of 3.3 and 5.5 ng/g ww. PFAS concentrations from the urban area were significantly higher than from background areas indicating that some of the species can be suitable as markers for PFAS emissions in an urban environment. Fieldfare eggs had surprisingly high concentrations of PFOS and PFCA concentrations from areas known to be or have been influenced by industry. Biota-soil-accumulation factor and magnification calculations indicate accumulation and magnification potential for several PFAS. Earthworm and fieldfare egg had average concentrations above the Canadian and European thresholds in diet for avian wildlife and predators. For earthworms, 18 % of the samples exceeded the European threshold (33 ng/g ww) of PFOS in prey for predators, and for fieldfare eggs, 35 % of the samples were above the same threshold. None of the soil samples exceeded a proposed PNEC of PFOS for soil living organisms of 373 ng/g dw.[2] |
| References |
[1]. Emerging and legacy per- and polyfluoroalkyl substances in water, sediment, and air of the Bohai Sea and its surrounding rivers. Environ Pollut. 2020 Aug;263(Pt A):114391. [2]. New insights from an eight-year study on per- and polyfluoroalkyl substances in an urban terrestrial ecosystem. Environ Pollut . 2024 Apr 15:347:123735. |
| Additional Infomation | The contamination of surface waters in China with Per- and polyfluoroalkyl (PFASs) has been extensively studied in recent decades, however, almost all studies have been conducted in small areas and/or limited samples, which are not representative of the nationwide contamination of surface water environments with PFASs. In this study, attempt was made to provide a comprehensive report about PFASs pollution in Chinese surface water based on the PRISMA. By analyzing 111 papers published between 2006 and 2022, we provide a systematic review of the pollution of PFASs in surface water environments in China. The results show that 26 PFASs contaminants were detected at least once in China's surface water environment and were mainly concentrated in the eastern part of China. Most surface water environments in China had mean PFASs concentrations below 100 ng/L. The most polluted place was the Xiaoqing River, where sampling results in 2020 showed PFASs concentrations as high as 25,429 ng/L, followed by the Tangxun Lake, the Xi River, the Daling River, the Majia River, the Baiyangdian Lake, the Liuxi River, the Jiaolai River, the Tuo River and the Zhimai River. The Xiaoqing River also has the highest concentration of the novel pollutant, with concentrations of HFPO-TA and HFPO-DA as high as 1039 ng/L and 164 ng/L. Based on the source analysis, fluoropolymer manufacturing plants are the main source of PFASs pollutants in surface water. The results of the base risk analysis using risk quotients value (RQ) method show that the RQ values of the Xiaoqing River, the surface water near Bohai Bay, the Majia River and the Tuo River PFOA are 36.9, 7.7, 3.6 and 2.1 respectively, which are high risk areas and require enhanced control. This study provides information on surface waters contaminated by PFASs nationwide, and the results can be used as a reference for the development of pollution control and management strategies for PFASs in surface waters in China.Ecotoxicol Environ Saf. 2023 Jun 23:262:115178. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3355 mL | 11.6776 mL | 23.3552 mL | |
| 5 mM | 0.4671 mL | 2.3355 mL | 4.6710 mL | |
| 10 mM | 0.2336 mL | 1.1678 mL | 2.3355 mL |