Physicochemical Properties
| Molecular Formula | C16H16F3N3 |
| Molecular Weight | 307.31355381012 |
| Exact Mass | 307.129 |
| Elemental Analysis | C, 62.53; H, 5.25; F, 18.55; N, 13.67 |
| CAS # | 1430208-73-3 |
| Related CAS # | 1430208-73-3 |
| PubChem CID | 71543346 |
| Appearance | White to off-white solid powder |
| LogP | 4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 22 |
| Complexity | 385 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | DVIJFJSZZNOTLP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H16F3N3/c1-10-7-11(2)9-12(8-10)21-15(20)22-14-6-4-3-5-13(14)16(17,18)19/h3-9H,1-2H3,(H3,20,21,22) |
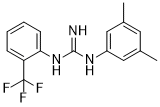
| Chemical Name | 1-(3,5-dimethylphenyl)-2-[2-(trifluoromethyl)phenyl]guanidine |
| Synonyms | 1A 116; 1A116; 1A-116 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | F3II (IC50 = 4 µM); MDA-MB-231 (IC50 = 21 µM); Rac1; Apoptosis |
| ln Vitro | 1A-116 (48 hours) has a concentration-dependent effect on the proliferation of MDA-MB-231 and F3II cells, with IC50 values of 4 μM and 21 μM, respectively [1]. 1A-116 (1, 10 μM; 12 h) dramatically increases Rac1 in F3II cells and, in a concentration-dependent way, decreases intracellular Rac1-GTP levels [1]. 1A-116 bursts the Rac1-P-Rex1 response at 50 and 100 μM over a 12-hour period [1]. LN229 cell proliferation is inhibited in a circadian manner by 1A-116 (20 μM; 5-hour interval over 25 h) [2]. At 10 HPS, 1A-116 (10 μM; 16 h) dramatically decreased cell migration, demonstrating temporal regulation. Hours post synchronization (HPS): Following serum shock, the elapsed time is measured in hours [2]. 1A-116 (20, 50 μM; 6 h) senses sterilization of cells in a manner that is dependent on the circadian rhythm [2]. At the GEF-Rac1 level, 1A-116 (100 nM) shows inhibitory Rac1 activity and decreases the thickness of the flipped layer mediated by Vav2 and Rac1, but not PAK1 [3]. 1][2] |
| ln Vivo | 1A-116 (intravenous; 3 mg/kg; once day for 21 days) showed strong anti-metastatic action without causing appreciable damage, with a 60% decrease in the total number of metastatic lung colonies in vivo. [1] 1A -116 (20 mg/kg; intraperitoneal; once daily; for ZT12, 73 days; for ZT3, 68 days) In tumor-bearing mice, treatment with ZT12 increased longevity in comparison to ZT3. Zeitgeber Time 12 (ZT12) denotes the time at which the lights go out (7 p.m. tonight), while ZT0 denotes the time at which they come on (7 a.m. tonight). [2]. Good wall availability is shown in 1A-116 [3]. |
| Cell Assay |
Cell Proliferation Assay[1][2] Cell Types: MDA-MB-231, F3II, LN229 Cell Tested Concentrations: 20 µM Incubation Duration: 48 hrs (hours); 5 hrs (hours) apart within 25 hrs (hours). Experimental Results: Inhibition of cell proliferation in a concentration-dependent and circadian manner. Cell viability assay [3] Cell Types: Ker-CT Human Keratinocytes with Oncogenic Vav2/Rac1 F28L/PAK1 Tyrosine 423 Tested Concentrations: 100 nM Incubation Duration: Experimental Results: Inhibition of Rac1 activity at the GEF-Rac1 level. Cell migration assay[2] Cell Types: LN229 Cell Tested Concentrations: 10 µM Incubation Duration: 16 hrs (hours) Experimental Results: Cell migration was diminished at 10 HPS, showing time dependence. Apoptosis analysis[2] Cell Types: LN229 Cell Tested Concentrations: 20, 50 µM Incubation Duration: 6 hrs (hours) Experimental Results: Apoptosis is induced in a circadian rhythm-dependent manner. Western Blot Analysis[1] Cell Types: F3II Cell Tested Concentrations: 1, 10 µM Incubation Duration: 12 hrs (hours) Experimental Results: Blocks Rac1-P-Rex1 interaction. Reduces intracellular Rac1-GTP levels in a concentration-dependent manner. |
| Animal Protocol |
Animal/Disease Models: Female BALB/c inbred mice (8 to 10 weeks old; average 20 g) [1] Doses: 3 mg/kg Route of Administration: intravenous (iv) (iv)injection; one time/day for 21 days. Experimental Results: demonstrated high antimetastatic activity. Animal/Disease Models: Male NIH Swiss FoxN1(Δ/Δ) nude mice (2 months old; GBM model) [2]. Doses: 20 mg/kg Route of Administration: intraperitoneal (ip) injection (ZT3, ZT12); one time/day, ZT12 for 73 days, ZT3 for 68 days. Experimental Results: ZT12-treated tumor-bearing mice survived longer compared with ZT3 treatment. |
| References |
[1]. Preclinical development of novel Rac1-GEF signaling inhibitors using a rational design approach in highly aggressive breast cancer cell lines. Anticancer Agents Med Chem. 2014;14(6):840-51. [2]. Timing of Novel Drug 1A-116 to Circadian Rhythms Improves Therapeutic Effects against Glioblastoma. Pharmaceutics. 2021 Jul 16;13(7):1091. [3]. Computational and in vitro Pharmacodynamics Characterization of 1A-116 Rac1 Inhibitor: Relevance of Trp56 in Its Biological Activity. Front Cell Dev Biol. 2020 Apr 15;8:240. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 61~100 mg/mL (198.5~325.4 mM) Ethanol: ~61 mg/mL (~198.5 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.14 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (8.14 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.14 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2540 mL | 16.2702 mL | 32.5404 mL | |
| 5 mM | 0.6508 mL | 3.2540 mL | 6.5081 mL | |
| 10 mM | 0.3254 mL | 1.6270 mL | 3.2540 mL |