Physicochemical Properties
| Molecular Formula | C30H46O4 |
| Molecular Weight | 470.68384 |
| Exact Mass | 470.339 |
| CAS # | 1449-05-4 |
| Related CAS # | 18β-Glycyrrhetinic acid;471-53-4 |
| PubChem CID | 10114 |
| Appearance | White to off-white solid powder |
| Density | 1.1±0.1 g/cm3 |
| Boiling Point | 588.3±50.0 °C at 760 mmHg |
| Melting Point | 331-333°C |
| Flash Point | 323.7±26.6 °C |
| Vapour Pressure | 0.0±3.7 mmHg at 25°C |
| Index of Refraction | 1.563 |
| LogP | 6.57 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 34 |
| Complexity | 965 |
| Defined Atom Stereocenter Count | 9 |
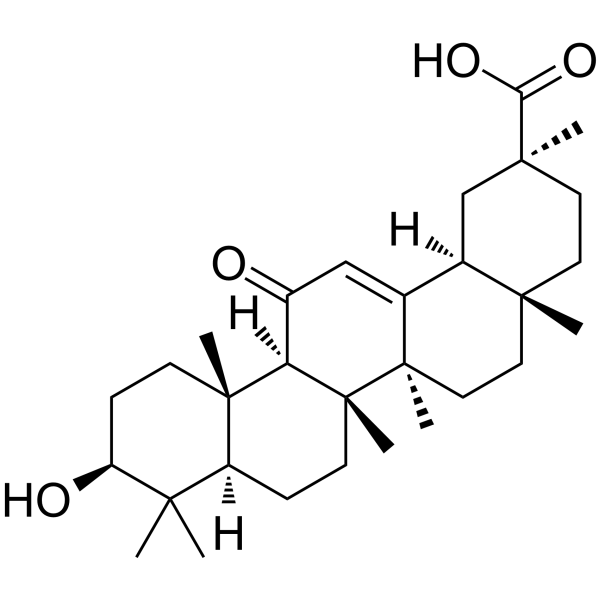
| SMILES | C[C@]12CC[C@](C[C@H]1C3=CC(=O)[C@@H]4[C@]5(CC[C@@H](C([C@@H]5CC[C@]4([C@@]3(CC2)C)C)(C)C)O)C)(C)C(=O)O |
| InChi Key | MPDGHEJMBKOTSU-YKLVYJNSSA-N |
| InChi Code | InChI=1S/C30H46O4/c1-25(2)21-8-11-30(7)23(28(21,5)10-9-22(25)32)20(31)16-18-19-17-27(4,24(33)34)13-12-26(19,3)14-15-29(18,30)6/h16,19,21-23,32H,8-15,17H2,1-7H3,(H,33,34)/t19-,21-,22-,23+,26+,27-,28-,29+,30+/m0/s1 |
| Chemical Name | (2S,4aS,6aR,6aS,6bR,8aR,10S,12aS,14bR)-10-hydroxy-2,4a,6a,6b,9,9,12a-heptamethyl-13-oxo-3,4,5,6,6a,7,8,8a,10,11,12,14b-dodecahydro-1H-picene-2-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | After 18α-glycyrrhetinic acid (18a-GA) therapy for 48 hours and 72 hours, the number of LX-2 cells was dramatically reduced by 14.8% and 31.2% correspondingly (P<0.05). Compared with the control group, after 48 and 72 hours of 18α-glycyrrhetinic acid administration, the proportion of LX-2 cells in the G0/G1 phase considerably increased, while the percentage of cells in the S phase decreased. 18α-Glycyrrhetinic acid increased cell apoptosis to 6.8% after 48 hours compared to the control group (2.5%), and at 72 hours, the percentage of apoptotic cells in LX-2 cells in the control and treatment groups were respectively 3.1% and 15.6% (P<0.01). In addition, 18α-glycyrrhetinic acid stimulates the expression of PPAR-γ and modifies several cell cycle and apoptosis-related proteins. 18α-Glycyrrhetinic acid also suppresses NF-κB DNA binding activity [1]. |
| ln Vitro |
After 18α-glycyrrhetinic acid (18a-GA) therapy for 48 hours and 72 hours, the number of LX-2 cells was dramatically reduced by 14.8% and 31.2% correspondingly (P<0.05). Compared with the control group, after 48 and 72 hours of 18α-glycyrrhetinic acid administration, the proportion of LX-2 cells in the G0/G1 phase considerably increased, while the percentage of cells in the S phase decreased. 18α-Glycyrrhetinic acid increased cell apoptosis to 6.8% after 48 hours compared to the control group (2.5%), and at 72 hours, the percentage of apoptotic cells in LX-2 cells in the control and treatment groups were respectively 3.1% and 15.6% (P<0.01). In addition, 18α-glycyrrhetinic acid stimulates the expression of PPAR-γ and modifies several cell cycle and apoptosis-related proteins. 18α-Glycyrrhetinic acid also suppresses NF-κB DNA binding activity [1]. 18α-Glycyrrhetinic acid (8 µM) treatment for 48 h and 72 h significantly reduced the number of human hepatic stellate cell line LX-2 by 14.8% and 31.2%, respectively, compared to the control group. A similar inhibitory effect was observed on rat cirrhotic fat-storing cells (CFSC). [1] Flow cytometric analysis showed that 18α-Glycyrrhetinic acid (8 µM) increased the percentage of LX-2 and CFSC cells in the G0/G1 phase and decreased the percentage in the S phase after 48 h and 72 h of treatment. [1] 18α-Glycyrrhetinic acid (8 µM) induced apoptosis in LX-2 cells, with apoptotic rates of 6.8% at 48 h and 15.6% at 72 h, compared to 2.5% and 3.1% in the control group, respectively. Similar pro-apoptotic effects were observed in CFSC cells. [1] Real-time PCR and Western blot analysis demonstrated that 18α-Glycyrrhetinic acid (8 µM) treatment increased the mRNA and protein levels of PPAR-γ in a time-dependent manner. [1] Western blot analysis revealed that 18α-Glycyrrhetinic acid (8 µM) decreased the protein level of cyclin D1 and increased the level of p27 in LX-2 cells. It also downregulated the anti-apoptotic proteins Bcl-xl and Bfl/A1, while upregulating the pro-apoptotic protein Bax. [1] A transcription factor activity assay showed that 18α-Glycyrrhetinic acid (8 µM) significantly inhibited NF-κB DNA-binding activity in LX-2 cells by 8% at 24 h and 17% at 48 h. [1] All the above effects of 18α-Glycyrrhetinic acid on cell proliferation, apoptosis, cell cycle, protein expression, and NF-κB activity were abolished by pre-treatment with the selective PPAR-γ antagonist GW9662 (0.5 µM). [1] |
| ln Vivo | 18α-glycyrrhetinic acid (18α-GA) treatment can greatly increase the longevity of nematode strains; 20 μg/mL is the most effective quantity. The outcomes demonstrated a considerable delay in the onset of paralysis symptoms following treatment with 18α-glycyrrhetinic acid. Treatment with 18α-glycyrrhetinic acid can also considerably lower Aβ deposition [2]. |
| Cell Assay |
Cell Viability and Proliferation Assay: Human LX-2 and rat CFSC cells were seeded in 96-well plates. After adherence, cells were treated with 18α-Glycyrrhetinic acid at concentrations ranging from 0 to 32 µM for 48 hours. Cell viability was assessed using a Cell Counting Kit-8 (CCK-8). Absorbance was measured at 450 nm. A concentration of 8 µM was selected for subsequent experiments as cell viability remained above 80%. [1] Cell Growth Determination: Cells were seeded in 24-well plates, starved for 24 hours, and then stimulated with medium containing 10% fetal bovine serum with or without 8 µM 18α-Glycyrrhetinic acid. Cell numbers were counted at 24, 48, and 72 hours post-treatment using a computer-equipped cell counter. [1] Cell Cycle Analysis by Flow Cytometry: Cells were seeded in 6-well plates, starved, and treated with 8 µM 18α-Glycyrrhetinic acid for 24, 48, and 72 hours. Cells were then harvested, and their DNA content was analyzed by flow cytometry to determine the distribution across different cell cycle phases (G0/G1, S, G2/M). [1] Apoptosis Analysis by Flow Cytometry: After treatment with 8 µM 18α-Glycyrrhetinic acid for specified times, cells were stained using an Annexin V-based staining kit according to the manufacturer's instructions. The percentage of apoptotic cells was quantified by flow cytometry. [1] Gene Expression Analysis (Real-time PCR): Total RNA was extracted from treated cells. Reverse transcription was performed. The mRNA levels of PPAR-γ were quantified by real-time PCR using SYBR Green, with GAPDH serving as the internal control. The fold change was calculated. [1] Protein Expression Analysis (Western Blot): Total protein was extracted from cultured cells. Proteins were separated by SDS-PAGE, transferred to membranes, and probed with specific primary antibodies against various targets (e.g., cyclins, p21, p27, PPAR-γ, Bcl-2 family proteins). GAPDH was used as a loading control. Protein bands were visualized using enhanced chemiluminescence. [1] NF-κB DNA-Binding Activity Assay: Nuclear proteins were extracted from treated cells. Equal amounts of nuclear protein were added to wells coated with an oligonucleotide containing the NF-κB consensus binding site. The binding of active NF-κB (p65 subunit) was detected using a specific primary antibody, followed by a horseradish peroxidase-conjugated secondary antibody, and quantified colorimetrically at 450 nm. [1] |
| Toxicity/Toxicokinetics |
Cytotoxicity: In a cell viability assay using CCK-8, treatment with 18α-Glycyrrhetinic acid at concentrations up to 16 µM for 48 hours resulted in cell viability above 80% for both LX-2 and CFSC cell lines. Higher concentrations may show increased toxicity. [1] |
| References |
[1]. 18α-glycyrrhetinic acid extracted from Glycyrrhiza radix inhibits proliferation and promotes apoptosis of the hepatic stellate cell line. J Dig Dis. 2013 Jun;14(6):328-36. [2]. 18α-Glycyrrhetinic Acid Proteasome Activator Decelerates Aging and Alzheimer's Disease Progression in Caenorhabditis elegans and Neuronal Cultures. Antioxid Redox Signal. 2016 Dec 1;25(16):855-869. |
| Additional Infomation |
Glycyrrhetinic acid is a pentacyclic triterpenoid that is olean-12-ene substituted by a hydroxy group at position 3, an oxo group at position 11 and a carboxy group at position 30. It has a role as an immunomodulator and a plant metabolite. It is a pentacyclic triterpenoid, a cyclic terpene ketone and a hydroxy monocarboxylic acid. It is a conjugate acid of a glycyrrhetinate. It derives from a hydride of an oleanane. Enoxolone (glycyrrhetic acid) has been investigated for the basic science of Apparent Mineralocorticoid Excess (AME). Enoxolone has been reported in Glycyrrhiza uralensis, Glycyrrhiza, and other organisms with data available. Enoxolone is a pentacyclic triterpenoid aglycone metabolite of glycyrrhizin, which is a product of the plant Glycyrrhiza glabra (licorice), with potential expectorant, and gastrokinetic activities. After administration, enoxolone inhibits the metabolism of prostaglandins by both 15-hydroxyprostaglandin dehydrogenase [NAD(+)] and prostaglandin reductase 2. Therefore, this agent potentiates the activity of prostaglandin E2 and F2alpha, which inhibits gastric secretion while stimulating pancreatic secretion and the secretion of intestinal and respiratory mucus, leading to increased intestinal motility and antitussive effects. Additionally, this agent inhibits 11 beta-hydroxysteroid dehydrogenase and other enzymes involved in the conversion of cortisol to cortisone in the kidneys. An oleanolic acid from GLYCYRRHIZA that has some antiallergic, antibacterial, and antiviral properties. It is used topically for allergic or infectious skin inflammation and orally for its aldosterone effects in electrolyte regulation. See also: Glycyrrhizin (is active moiety of); Ammonium Glycyrrhizate (is active moiety of); Glycyrrhiza Glabra (part of). 18α-Glycyrrhetinic acid is an epimer of 18β-glycyrrhetinic acid at the 18-H position. It is described as targeting the liver more strongly and having a stronger protective effect on hepatocytes with lower toxicity compared to 18β-glycyrrhetinic acid. [1] The study suggests that the anti-proliferative and pro-apoptotic effects of 18α-Glycyrrhetinic acid on hepatic stellate cells are mediated, at least in part, through the upregulation of PPAR-γ expression and the subsequent inhibition of NF-κB activity. This highlights its potential as a candidate for anti-fibrotic therapy in liver fibrosis. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~11.36 mg/mL (~24.14 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 1.14 mg/mL (2.42 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 11.4 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1.14 mg/mL (2.42 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 11.4 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1246 mL | 10.6229 mL | 21.2459 mL | |
| 5 mM | 0.4249 mL | 2.1246 mL | 4.2492 mL | |
| 10 mM | 0.2125 mL | 1.0623 mL | 2.1246 mL |