Physicochemical Properties
| Molecular Weight | 589.653650283813 |
| CAS # | 141553-69-7 |
| Related CAS # | 1425501-12-7 (free acid) |
| Appearance | Typically exists as solids (or liquids in special cases) at room temperature |
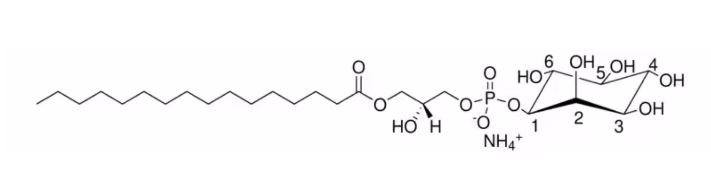
| SMILES | O(C1C(C(O)C(O)C(O)C1O)O)P(O)(=O)OCC(O)COC(=O)CCCCCCCCCCCCCCC.N |
| Chemical Name | 1-hexadecanoyl-2-hydroxy-sn-glycero-3-phospho-(1′-myo-inositol) (ammonium salt) |
| Synonyms | 16:0 Lyso PI ammonium salt; 1-palmitoyl-2-hydroxy-sn-glycero-3-phosphoinositol (ammonium salt); 141553-69-7 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Phosphoglycerides |
| ln Vitro | Lysophosphatidylinositol (LPI) is a well-known bioactive lipid that is able to activate signalling cascades relevant to cell proliferation, migration, survival and tumorigenesis. Our previous work suggested that LPI is involved in cancer progression since it can be released in the medium of Ras-transformed fibroblasts and can function as an autocrine modulator of cell growth. Different research groups have established that LPI is the specific and functional ligand for G-protein-coupled receptor 55 (GPR55) and that this GPR55-LPI axis is able to activate signalling cascades that are relevant for different cell functions. Work in our laboratory has recently unravelled an autocrine loop, by which LPI synthesized by cytosolic phospholipase A₂ (cPLA₂) is pumped out of the cell by ATP-binding cassette (ABC) transporter C1 (ABCC1)/multidrug resistance protein 1 (MRP1), initiating a signalling cascade downstream of GPR55. Our current work suggests that blockade of this pathway may represent a novel strategy to inhibit cancer cell proliferation[1]. |
| ln Vivo | Recent studies have identified a genetic variant rs641738 near two genes encoding membrane bound O-acyltransferase domain-containing 7 (MBOAT7) and transmembrane channel-like 4 (TMC4) that associate with increased risk of non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), alcohol-related cirrhosis, and liver fibrosis in those infected with viral hepatitis (Buch et al., 2015; Mancina et al., 2016; Luukkonen et al., 2016; Thabet et al., 2016; Viitasalo et al., 2016; Krawczyk et al., 2017; Thabet et al., 2017). Based on hepatic expression quantitative trait loci analysis, it has been suggested that MBOAT7 loss of function promotes liver disease progression (Buch et al., 2015; Mancina et al., 2016; Luukkonen et al., 2016; Thabet et al., 2016; Viitasalo et al., 2016; Krawczyk et al., 2017; Thabet et al., 2017), but this has never been formally tested. Here we show that Mboat7 loss, but not Tmc4, in mice is sufficient to promote the progression of NAFLD in the setting of high fat diet. Mboat7 loss of function is associated with accumulation of its substrate lysophosphatidylinositol (LPI) lipids, and direct administration of LPI promotes hepatic inflammatory and fibrotic transcriptional changes in an Mboat7-dependent manner. These studies reveal a novel role for MBOAT7-driven acylation of LPI lipids in suppressing the progression of NAFLD[2]. |
| Enzyme Assay |
Lysophosphatidylinositol acyltransferase biochemical assay[2] [1-14C]-arachidonyl-CoA was obtained commercially. Lysophosphatidylinositol (LPI) substrates (16:0 LPI, 18:0 LPI, and 18:1 LPI) and lipid standards used in the enzyme assays were obtained commercially. Liver microsomes isolated from Mboat7 ASO and control ASO treated mice on both chow and high fat diet was used to measure LPIAT activity. The assay buffer contained 50 mM Tris-HCL (pH 8.0), 150 mM NaCl, 50 μM 18:0-LPI, 20 μM [1-14C]arachidonyl-CoA (0.025 µCi), and 15 µg of the microsomes in a total volume of 100 µL. Substrate was prepared in CHAPS (0.01 mM final concentration). The assay mixture was incubated for 30 min at 37°C, and the reaction was stopped by the addition of 1:2:1 (v/v/v) chloroform:methanol:2% orthophosphoric acid. The lipids were extracted and separated on a silica–TLC plate using chloroform/acetone/acetic acid/methanol/water (50:20:15:10:5, v/v) as the solvent system. The individual lipid molecules were identified by migration with respect to standards. Enzymatic products were monitored by autoradiogram, corresponding spots were scraped from the TLC plate, and the radioactivity was quantified with a liquid scintillation counter. |
| Animal Protocol |
Isolation and characterization of lipid droplets from mouse liver[2] Hepatic LDs were isolated by sucrose gradient centrifugation as we have previously described (Ferguson et al., 2017). Approximately 100 mg of tissue was minced with a razor blade on a cold surface. Minced tissue was transferred to a Potter-Elvehjem homogenizer, and then 200 μl of 60% sucrose was added to the tissue sample and incubated on ice for 10 min. Next, 800 μl of lysis buffer was added and mixed, and then incubated on ice for 10 min. Samples were homogenized with five strokes of a Teflon pestle and transferred to a 2 ml centrifuge tube. Lysis buffer (600 μl) was carefully layered on top of homogenate and centrifuged for 2 hr at 20,000 g at 4°C. The tube was then frozen at −80°C and cut at the 1,000 μl mark. The bottom piece of the centrifuge tube contained the non-LD fraction, which was allowed to thaw before being transferred to a new tube. The LD fraction was collected by cutting an ∼4–6 mm piece from the top of the ice cylinder and placing it in a new 2 ml tube. To increase the purity of the LD fraction, this process was repeated once more. Briefly, 200 μl of 60% sucrose was added to the LD fraction. Next, 800 μl of lysis buffer was added and mixed followed by careful layering with 600 μl of lysis buffer and then centrifugation for 2 hr at 20,000 g at 4°C. After freezing at −80°C, the tube was cut and the LD fraction was collected by cutting an ∼4–6 mm piece from the top of the ice cylinder and placing it in a new tube. Protein analysis was performed using the modified Lowry assay, as previously described (Ferguson et al., 2017), and Western blotting was performed as described below. MBOAT7 substrate (lysophosphatidylinositol) and product (phosphatidylinositol) lipids in the LD fraction were extracted and quantified using the targeted LC-MS/MS methods described below. |
| References |
[1]. Lysophosphatidylinositol: a novel link between ABC transporters and G-protein-coupled receptors. Biochemical Society transactions, 2014, 42(5), 1372-1377. [2]. Obesity-linked suppression of membrane-bound O-acyltransferase 7 (MBOAT7) drives non-alcoholic fatty liver disease. eLife, 2019 Oct 17:8:e49882. |
| Additional Infomation |
16:0 Lyso PI (1-palmitoyl-2-hydroxy-sn-glycero-3-phosphoinositol) has been used as a lipid standard in the optimization of the liquid chromatography–mass spectrometry (LC-MS) method conditions and also has been used as lysophosphatidylinositol (LPI) substrates in lysophosphatidylinositol acyltransferase biochemical assay to assay lyso-phosphatidylinositol acyltransferase (LPIAT) activity of liver microsomes. It may also be used as synthetic LPI to compare its effect with LPI from liver in transferrin (Tf)mediated endocytosis. Increased circulating levels of L-alpha-lysophosphatidylinositol (LPI) are associated with cancer and LPI is a potent ligand for the G-protein-coupled receptor GPR55. Lysophosphatidylinositol (LPI) has the ability to modulate cancer cell migration. It can control the functions of endothelium. LPI can serve as an autocrine regulator of cell growth, hence its known to participate in cancer progression. It can stimulate signaling cascades, that are essential for survival, migration and proliferation of cells. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.6959 mL | 8.4796 mL | 16.9592 mL | |
| 5 mM | 0.3392 mL | 1.6959 mL | 3.3918 mL | |
| 10 mM | 0.1696 mL | 0.8480 mL | 1.6959 mL |