|
15(S)-Latanoprost is a latanoprost analog in which the hydroxyl at carbon 15 is inverted relative to latanoprost. The IC50 values for the free acid forms of latanoprost and 15(S)-latanoprost were determined to be 3.6 nM and 24 nM, respectively, in a FP receptor binding assay using the cat iris sphincter muscle. A 3 μg dose of 15(S)-latanoprost caused a 1 mmHg reduction of IOP in normotensive cynomolgus monkeys.
|
Physicochemical Properties
| Molecular Formula |
C26H40O5
|
| Molecular Weight |
432.5928
|
| Exact Mass |
430.271
|
| CAS # |
145773-22-4
|
| PubChem CID |
10477939
|
| Appearance |
Typically exists as solid at room temperature
|
| Density |
1.1±0.1 g/cm3
|
| Boiling Point |
573.4±50.0 °C at 760 mmHg
|
| Flash Point |
185.0±23.6 °C
|
| Vapour Pressure |
0.0±1.7 mmHg at 25°C
|
| Index of Refraction |
1.579
|
| LogP |
3.56
|
| Hydrogen Bond Donor Count |
3
|
| Hydrogen Bond Acceptor Count |
5
|
| Rotatable Bond Count |
14
|
| Heavy Atom Count |
31
|
| Complexity |
526
|
| Defined Atom Stereocenter Count |
5
|
| SMILES |
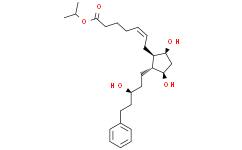
CC(C)OC(=O)CCC/C=C\C[C@@H]1[C@@H](CC[C@@H](CCC2=CC=CC=C2)O)[C@@H](C[C@@H]1O)O
|
| InChi Key |
GGXICVAJURFBLW-SCTZCWPJSA-N
|
| InChi Code |
InChI=1S/C26H40O5/c1-19(2)31-26(30)13-9-4-3-8-12-22-23(25(29)18-24(22)28)17-16-21(27)15-14-20-10-6-5-7-11-20/h3,5-8,10-11,19,21-25,27-29H,4,9,12-18H2,1-2H3/b8-3-/t21-,22-,23-,24+,25-/m1/s1
|
| Chemical Name |
propan-2-yl (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(3S)-3-hydroxy-5-phenylpentyl]cyclopentyl]hept-5-enoate
|
| Synonyms |
15(S)-Latanoprost; 145773-22-4; (15S)-Latanoprost; 15-epi-Latanoprost; Latanoprost, (15S)-; P840K2J2DL; propan-2-yl (Z)-7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(3S)-3-hydroxy-5-phenylpentyl]cyclopentyl]hept-5-enoate; 5-Heptenoic acid, 7-[(1R,2R,3R,5S)-3,5-dihydroxy-2-[(3S)-3-hydroxy-5-phenylpentyl]cyclopentyl]-, 1-methylethyl ester, (5Z)-;
|
| HS Tariff Code |
2934.99.9001
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
|
Biological Activity
| Targets |
FP Receptor
|
| ln Vitro |
In addition, the activity of the analogues on FP receptors was studied in vitro[1].
|
| ln Vivo |
A series of phenyl-substituted analogues of prostaglandin F2 alpha (PGF2 alpha) were prepared and evaluated for ocular hypotensive effect and side effects in different animal models. In addition, the activity of the analogues on FP receptors was studied in vitro. The results were compared with those of PGF2 alpha and its isopropyl ester. The phenyl-substituted PGF2 alpha analogues exhibited good intraocular pressure reducing effect, were more selective, and exhibited a much higher therapeutic index in the eye than PGF2 alpha or its isopropyl ester. The analogues exhibited high activity on FP receptors in a stereoselective manner for the 15 alpha-hydroxyl group[1].
|
| References |
[1]. Phenyl-substituted prostaglandins: Potent and selective antiglaucoma agents. J. Med. Chem. 36(2), 243-248 (1993).
|
|
Solubility Data
| Solubility (In Vitro) |
May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples
|
| Preparing Stock Solutions |
|
1 mg |
5 mg |
10 mg |
| 1 mM |
2.3117 mL |
11.5583 mL |
23.1166 mL |
| 5 mM |
0.4623 mL |
2.3117 mL |
4.6233 mL |
| 10 mM |
0.2312 mL |
1.1558 mL |
2.3117 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles. |