Physicochemical Properties
| Molecular Formula | C12H8 |
| Molecular Weight | 152.19192 |
| Exact Mass | 152.063 |
| CAS # | 15727-65-8 |
| Related CAS # | 31961-12-3 |
| PubChem CID | 123357 |
| Appearance | Colorless to light yellow liquid |
| Density | 1.070 g/mL at 25ºC(lit.) |
| Boiling Point | 270.4ºC at 760 mmHg |
| Flash Point | 106.3ºC |
| Vapour Pressure | 0.0114mmHg at 25°C |
| Index of Refraction | n20/D 1.6500(lit.) |
| LogP | 2.821 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 0 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 12 |
| Complexity | 195 |
| Defined Atom Stereocenter Count | 0 |
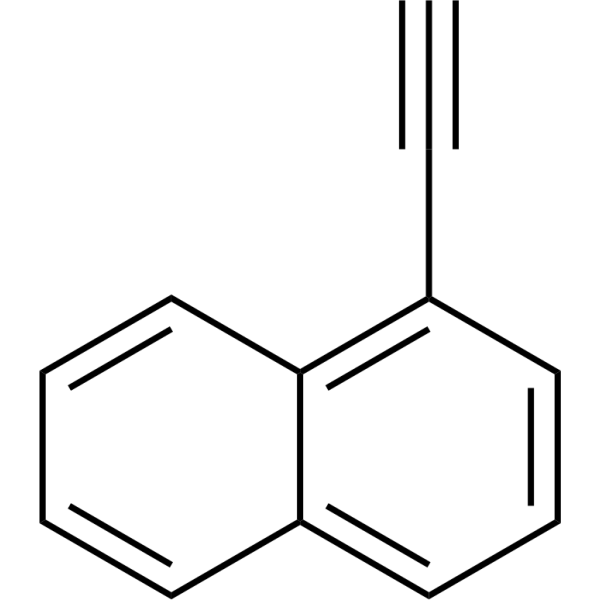
| SMILES | C#CC1=C2C=CC=CC2=CC=C1 |
| InChi Key | MCZUXEWWARACSP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C12H8/c1-2-10-7-5-8-11-6-3-4-9-12(10)11/h1,3-9H |
| Chemical Name | 1-ethynylnaphthalene |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | 1-Ethynylnaphthalene is a selective inhibitor of cytochrome P450 IB1. It does not effect 2-hydroxylation but inhibits 4-hydroxylation by 38%. At higher doses, 1-ethynylnaphthalene can inhibit the 2-hydroxylation of estradiol (E2) 2-hydroxylation by roughly 30% and the 4-hydroxylation of E2 by up to 80% [1]. |
| ln Vitro |
1-Ethynylnaphthalene is a selective inhibitor of cytochrome P450 IB1. It does not effect 2-hydroxylation but inhibits 4-hydroxylation by 38%. At higher doses, 1-ethynylnaphthalene can inhibit the 2-hydroxylation of estradiol (E2) 2-hydroxylation by roughly 30% and the 4-hydroxylation of E2 by up to 80% [1]. 1-Ethynylnaphthalene was used as a tool inhibitor to study estrogen metabolism. In incubations with human uterine myoma microsomes and 5 μM estradiol (E₂), 10 μM 1-Ethynylnaphthalene did not affect the 2-hydroxylation of E₂ (97% of control activity) but inhibited the 4-hydroxylation of E₂ by 38% (62% of control activity). [1] In human uterine myometrium microsomes, 1-Ethynylnaphthalene (at an unspecified concentration, likely similar) inhibited 4-hydroxylation by less than 25% and did not affect 2-hydroxylation. [1] At higher concentrations (concentration not specified), 1-Ethynylnaphthalene inhibited the 2-hydroxylation of E₂ by approximately 30% and the 4-hydroxylation of E₂ by up to 80%. [1] |
| Enzyme Assay |
The study utilized a microsome-mediated catechol estrogen formation assay. Briefly, microsomal protein (750–1500 µg) was incubated with 5 mM NADPH and 1–100 µM [³H]estradiol as substrate in a Tris-HCl/Hepes buffer (pH 7.4) containing 5 mM ascorbic acid at 30°C for 30 minutes. Reactions were terminated by rapid freezing. Trace amounts of ¹⁴C-labeled catechol estrogens were added to correct for losses. Metabolites were adsorbed onto neutral alumina, washed, eluted with HCl, and separated by thin-layer chromatography. Inhibitors like 1-Ethynylnaphthalene were dissolved in ethanol (2 µL) and added to the incubation mixture. [1] |
| References |
[1]. 4-Hydroxylation of estradiol by human uterine myometrium and myoma microsomes: implications for the mechanism of uterine tumorigenesis. Proc Natl Acad Sci U S A. 1995 Sep 26;92(20):9220-4. |
| Additional Infomation |
1-Ethynylnaphthalene is categorized as an ethynylated polycyclic hydrocarbon inhibitor. [1] It is used as a selective chemical probe to distinguish between different cytochrome P450 enzymes involved in estrogen metabolism, specifically to inhibit the P450 IB1-related activity responsible for estradiol 4-hydroxylation. [1] The study suggests that the 4-hydroxylase activity in human uterine tissues is catalyzed by a form(s) of cytochrome P450 related to P450 IB1, based partly on the inhibitory profile of compounds like 1-Ethynylnaphthalene. [1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 125 mg/mL (~821.34 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (13.67 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (13.67 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (13.67 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.5707 mL | 32.8537 mL | 65.7073 mL | |
| 5 mM | 1.3141 mL | 6.5707 mL | 13.1415 mL | |
| 10 mM | 0.6571 mL | 3.2854 mL | 6.5707 mL |